Surface characterization of calcium phosphate coating formed on chitosan and alkali-treated titanium metal
Abstract
The calcium phosphate coating on various pretreated metals was prepared by soaking in modified simulated body fluid (m-SBF) solution. The coating structure and its surface morphologies were determined by x-ray diffraction, Fourier transform infrared spectroscopy, and scanning electron microscopy. The results revealed significant differences in morphology and composition of the calcium phosphate coatings with and without chitosan and NaOH-pretreated commercially pure titanium (cp-Ti) substrate. The calcium phosphates formed on chitosan coated-Ti pretreated with NaOH were ~ 350 nm-sized resulting in strong bonding of the apatite layer with the substrates and a uniform gradient of stress transfer from coating materials to the Ti-substrate. After NaOH pretreatment, the hydroxyl groups bind to Ca2+ to attract PO43- anions, eventually resulting in a continuous layer of calcium phosphate on chitosan coated-Ti substrate during immersion in m-SBF solution. The chitosan coated-Ti showed hydrophobic surface while NaOH pretreatment resulted in maximum hydrophilicity to the Ti substrate. Due to improved wettability of Ti by NaOH pretreatment before chitosan coating, aggregation of calcium phosphate was prevented and size-controlled composite materials were obtained.
초록
본 연구에서는 다양하게 전처리된 금속을 생체유사용액(m-SBF)에 침적하여 표면에 인산칼슘 층을 형성하였다. 코팅층의 구조와 표면 형상을 X-선 회절 분석기(XRD), 퓨리에 변환 적외선 분광분석기(FT-IR) 및 주사전자현미경(SEM)을 이용하여 분석하였다. 상용 순 타이타늄(cp-Ti) 기판에 대한 키토산 및 NaOH 전처리 시행 유무에 따라 형성되는 인산칼슘코팅 층의 형태 및 구조가 크게 차이를 보였다. NaOH로 전처리 한 후 키토산을 코팅한 타이타늄 상에 형성된 인산칼슘은약 350nm 크기이었고, 코팅 물질에서 타이타늄 기판으로의 균일한 응력 전달 구배로 아파타이트 층과 기판이 강한결합을 보였다. NaOH 전처리에 의해 생성된 표면의 경우, 생체유사용액에 침적되면 수산기에 Ca2+가 결합하여 PO43-을 끌어와서 키토산 코팅된 Ti 기판 상에 인산칼슘 층이 연속적으로 생성되었다. NaOH 전처리에 의한 타이타늄 표면은높은 친수성을 보였지만, 키토산 코팅된 타이타늄은 소수성 표면을 보였다. NaOH 전처리 후 키토산이 코팅된 경우타이타늄 기판의 젖음성이 향상되어 생성된 인산칼슘 층이 서로 응집되는 것을 막아 입자 크기가 조절된 복합 코팅층을얻을 수 있었다.
Keywords:
Calcium phosphate, Chitosan, Sodium hydroxide pretreatment, Modified simulated body fluid (m-SBF), Mineralization키워드:
인산칼슘, 키토산, 수산화나트륨 전처리, 생체유사용액, 미네랄Introduction
Calcium phosphate has been used as a biologically active layer for metallic implants (1-3). The metallic implants exhibit high corrosion resistance and outstanding mechanical properties but lack chemical integration with bone. Various surface modifications have been investigated in the past decades to induce bone bioactivity on the metal surface (4-7). Chemical treatment of titanium is a simple and effective technique implemented widely in recent years. The reagents most frequently employed in this type of treatment include NaOH (8) and H2O2 (9). Treatment with NaOH solution results in sodium titanate gel layer on the surface of Ti whereas H2O2 produces a titania gel layer. Both gel layers are considered bioactive as they induce deposition of bone-like apatite during soaking in simulated body fluid (SBF).
Bone-like apatite was formed on the surface of pure titanium pretreated with NaOH solution after immersion in SBF. A sodium titanate gel layer formed by NaOH pretreatment induces formation of bone-like apatite during immersion in SBF. The gel layer initiates apatite nucleation per se, followed by spontaneous growth in the presence of calcium and phosphate ions. Chitosan (CS) is one of the most promising biopolymers for tissue engineering and orthopedics and CS-based materials attracted considerable attention (10). Calcium phosphate-based composite materials are usually generated as a bioactive layer to initiate the formation of hydroxyapatite (HA) layer (11-13). The chitosan/calcium phosphate composites have been fabricated by various methods, such as sol-gel techniques (14), dip coating (15), plasma spray (16) and other methods. Many methods of pre-treatment of titanium surface before immersion in SBF solution have been widely studied, including sandblasting (17), alkali/acid etching (18), heat treatment (19) or combination of these three approaches (20).
This study investigated the bone-like apatite formation induced by Ti metal before and after pretreatment with chitosan (CS) and alkali. We studied the altered composition and morphology of calcium phosphate (CaP) coating in response to chitosan and NaOH pretreatment. All the samples were immersed in modified simulated body fluid (m-SBF) for 3 days to induce the formation of bone-like apatite layer. The surface morphology, chemical structure, and wettability were characterized using field emission electron microscopy (FE-SEM)/energy-dispersive X-ray spectroscopy (EDS), X-ray diffractometer (XRD), Fourier transform infrared spectroscopy (FT-IR), and contact angle analysis.
Materials and Methods
1. Preparation of substrate metal and treatment solution
Commercially pure titanium (cp-Ti) of ASTM Grade 2 (Daito Steel Co. Ltd., Japan) was used as a substrate metal. It was cut to a disk measuring 10 ㎜ in diameter and 1 ㎜ in thickness. The disk was degreased with acetone and water, polished with up to #2000 SiC paper and ultrasonically cleaned in ethanol and distilled water. Chitosan (CS) flake with a molecular weight of about 520,000 g mol-1 and 85 % degree of deacetylation (Sehwa Co., Korea) was used. Chitosan solution was prepared by dissolving chitosan (1 g) into 1 % acetic acid (100 mL) with stirring for 5 h to obtain a perfectly transparent solution. All the chemicals were purchased from Aldrich Chemical Corporation and used without further purification.
2. Treatment of titanium substrate and immersion in m-SBF
A 20 nm thick chitosan layer was obtained by spin coating with 1% chitosan solution on the cp-Ti substrate at a spin speed of 5,000 rpm for 20 s. The films were dried at 40 ℃ for 20 min in a dry oven and the spin coating was performed twice. The m-SBF containing higher concentrations of inorganic ions compared with that of Kokubo’s SBF solution was used to fabricate the precipitated calcium phosphate as powders. The m-SBF used in this study was prepared according to the methods reported previously (21). The m-SBF solution was prepared by doubling the concentration of CaCl2 and KH2PO4 compared with that of SBF solution. In brief, the composition of SBF solution in distilled water was as follows: 141 mM NaCl, 4.0 mM KCl, 0.5 mM MgSO4, 1.0 mM MgCl2, 4.2 mM NaHCO3, 2.5 mM CaCl2ㆍ2H2O, and 1.0 mM KH2PO4. The m-SBF solution was incubated in an electric furnace at 37 ℃ for 3 days and its pH was adjusted to 7.4 at 37 ℃. The cp-Ti substrates were soaked in 100 ml of 5.0 M NaOH aqueous solutions for 2h, and the samples were rinsed with distilled water and dried in air (22).
3. Surface characterization of the samples
The morphology of the samples was studied using field emission electron microscopy (FE-SEM, Hitachi-S4700, Japan). The chemical composition and phase purity of the coatings were characterized using energy-dispersive X-ray spectroscopy (EDS, RMAX energy EX-200, Horiba, Japan) and X-ray diffractometer (XRD, PANalytical, X'Pert PRO, Netherlands) with Cu Kα radiation (30mA, 40kV). The functional groups were confirmed by Fourier transform infrared spectroscopy (Spectrum 400, PerkinElmer, UK) with an attenuated total reflectance (ATR) accessory. The contact angle was measured to evaluate the wettability of the cp-Ti specimens before and after the various treatments. An equal volume of distilled water was added to each sample using a micropipette, and the shape of the drops or surface spread was measured in terms of contact angle.
Results and Discussion
1. Surface morphology and chemical composition of treated sample surfaces
The surface SEM micrographs of untreated cp-Ti substrates showed a few grinding marks without any porosity (Figure 1a) compared with cp-Ti substrates soaked in 5.0 M NaOH solution at 80 ℃ for 2h, which revealed porous network structures (Figure 1b). During alkali treatment, the Ti-O passive layer dissolves to form sodium titanate (NaxH2-xTi3O7). The amorphous sodium titanate layer formed on the surface induces calcium phosphate nucleation (23).
The SEM images following CaP coating of pretreated substrates after incubation in m-SBF at 37 ℃ for 3 days are presented in Figure 2. The nucleation of calcium phosphate crystals took place first, and the crystals grew on top of the cp-Ti surface by immersion in m-SBF (Figure 2a). Accumulation of calcium phosphate on the surface was initiated as small particles following by clusters. The CaP coating of NaOH-treated cp-Ti (Figure 2b) yielded larger globules. In Figure. 2c, the bio-mimetic coating onto the surface of chitosan spin-coated on NaOH-pretreated cp-Ti yielded spherical aggregates of nanoparticles. As shown in Figure. 2d, the agglomerated sphere-like crystallites varied in size after 3 days of immersion on chitosan-coated cp-Ti.

FE-SEM images of CaP/CS composites incubated in m-SBF for 3 days with (a) cp-Ti, (b) NaOH-treated cp-Ti, (c) chitosan spin-coated on NaOH-pretreated cp-Ti, and (d) chitosan-spin-coated cp-Ti substrates. Corresponding figures at high magnification of the squared areas showing low magnification images on the left side are also included.
The aggregates of island-like spheroidal particles on the surfaces of chitosan-coated Ti substrates were much larger than those of spheroidal particles on chitosan spin-coated surfaces of NaOH-pretreated substrates. A decent Ti-OH layer is vital to the nucleation of calcium phosphate with minimal aggregation. Upon exposure to m-SBF, the Ca2+ and Mg2+ ions from m-SBF adsorb on to the NaOH-pretreated substrates. The adsorbed Ca2+ and Mg2+ ions accelerate calcium phosphate nucleation.
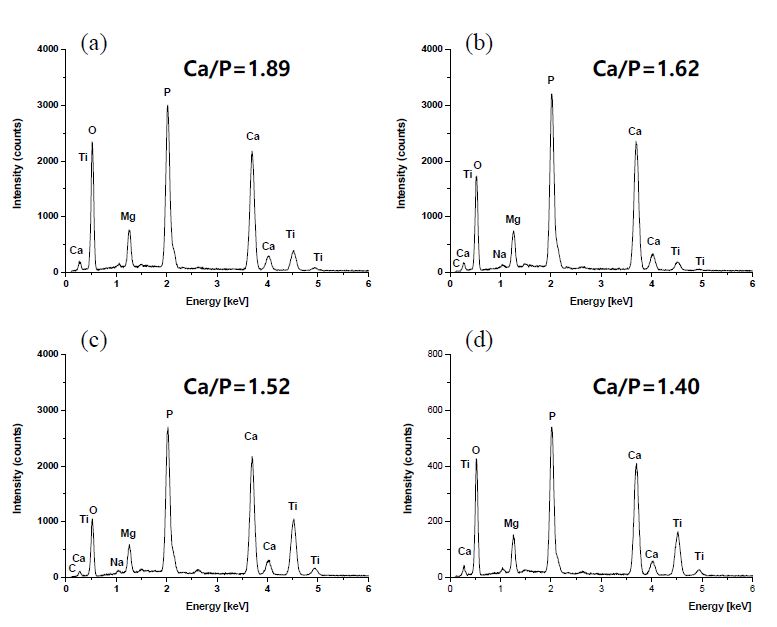
In EDS analysis, sharp peaks related to calcium and phosphorus confirmed the formation of CaP phases on all surfaces with a Ca/P ratio ranging from 1.40 to 1.89. Depending on the different CaP coating thickness of the layers, peaks of different intensities related to Ti were also observed (Figure 3). Furthermore, additional peaks corresponding to C and Mg were also detected. The C peak was either due to chitosan or carbonate ions in the CaP/CS coating. The Mg2+ ions may partially substitute Ca2+ ions in the CaP lattice structure. Mg ions act similar to a growth factor during the early stages of osteogenesis and promote bone formation. Mg ions play an important role in the biological activity, and interaction between bone mineral and calcium phosphate-based implant materials by influencing crystal growth, surface chemistry, and morphology (24). The Ca/P ratio decreased significantly when the Ti substrate was coated with CS prior to HA mineralization.
2. Chemical characterization of specimens
FT-IR was conducted to determine the chemical formula of the CaP coating layer formed on the treated substrates (Figure 4). The bands at approximately 1,016 cm−1 to 1,026 cm−1 are due to P–O and P=O stretching of the phosphate group, respectively. The symmetric and asymmetric vibrations of PO43− at 1,015 – 1,120 cm−1 and 873 cm−1 confirmed apatite peaks of CaP layer on all the substrates (25). The peaks of C-O asymmetric stretching at 1,415– 1,490 cm−1 were too high to correspond to the apatite, which suggests similarity with those usually observed for the carbonated apatite (i.e., substitution of OH− by CO32− ions). The bands at 602 cm−1 and 3,291 cm−1 are attributed to the structural OH groups in the HAP crystals. The absorption peaks around the wave numbers of 1,557 cm−1 and 1,400 cm−1 corresponded to the bending vibrations of N-H and C-H from chitosan, respectively. The peak intensity of P-OH group at 992 cm−1 and 1,016 cm-1 in CaP and CaP/CS coating on NaOH-pretreated substrate (i.e., Na-Ti and Chi-Na-Ti) decreased. The decreased peak intensity of P-OH groups with CaP is attributed to the OH of NaOH occupying the binding site.
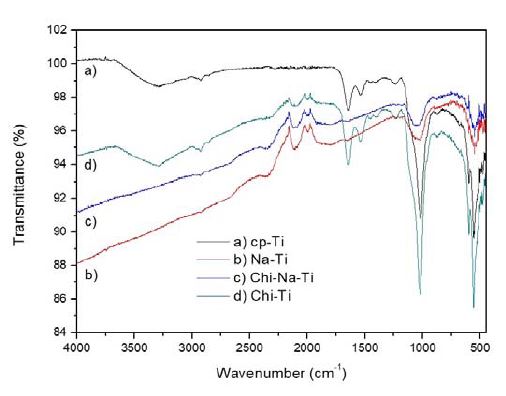
FT-IR spectra of CaP/CS composites after incubation in m-SBF at 37 ℃ for 3 days with (a) cp-Ti (cp-Ti), (b) NaOH-treated cp-Ti (Na-Ti), (c) chitosan spin-coated on NaOH pretreated cp-Ti (Chi-Na-Ti), and (d) chitosan-spin-coated cp-Ti (Chi-Ti) substrates.
The absence of the apatite OH stretching peaks at 602 cm−1 also partly demonstrates the substitution of carbonate ions. The characteristic peak of the apatite at 1,535 cm−1 was not observed with NaOH-pretreated substrate. The peaks with cp-Ti and Chi-Ti substrates were consistent with previous investigations confirming apatite formation after immersion in m-SBF.
3. Crystalline structure and wettability of specimens
A broad bump in the XRD patterns of cp-Ti treated with NaOH solution (i.e., Na-Ti and Chi-Na-Ti) suggests that the network structure on the surface is an amorphous sodium titanate hydrogel. Compared with the pattern of untreated cp-Ti substrate, a broad bump and small peaks were observed at around 24°, 28° and 48° (Figure 5b) indicating formation of an amorphous sodium titanate phase by NaOH treatment (26,27). All substrates showed peaks at 2θ = 35, 38.2, 40.1 and 52.7°, which were related to (100), (002), (101) and (102) planes of Ti, respectively. The XRD patterns of cp-Ti and Chi-Ti showed characteristic peaks of HA at 2θ = 25.9 and 31.8°. The peak at 31° appearing only in the XRD patterns of Na-Ti and Chi-Na-Ti was attributed to sodium titanate (Na2Ti5O11).
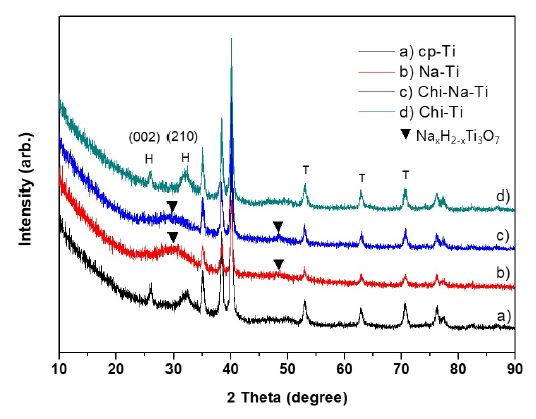
XRD diffraction patterns of CaP/CS composites after incubation in m-SBF at 37℃ for 3 days with (a) cp-Ti, (b) Na-Ti, (c) Chi-Na-Ti, and (d) Chi-Ti substrates.
Surface wettability is one of the most important parameters affecting the biological response of implanted materials. Figure 6 showed the typical shape of water drop on Ti substrates before and after alkali pretreatment. The average water contact-angles for the Chi-Ti samples were 75.5 ± 1.6°, which indicated the chitosan-coated cp-Ti surface was slightly hydrophobic. However, water dropped onto alkali-treated Ti (Na-Ti) surface formed a 10.4 ± 0.7° contact angle showing its hydrophilicity (28). This super-hydrophilicity of alkali-treated surfaces ensures uniform HA/CS coating of Ti substrates (Chi-Na-Ti, 52.5 ± 3.6°), which may enhance the biocompatibility of CS coatings compared to cp-Ti of 67.1 ± 2.8°.
Conclusions
Calcium phosphate coating was successfully synthesized on variously treated Ti-substrates by incubating in m-SBF. The chitosan binds calcium phosphate compound with the Ti-substrate and controls the size of calcium phosphate formation in m-SBF. Spontaneously formed calcium phosphate layer was tightly bonded to the substrates after NaOH pretreatment. The chitosan coated-Ti resulted in a hydrophobic surface while NaOH pretreatment generated the highest hydrophilicity on the Ti substrate. Due to the improved wettability of Ti by NaOH pretreatment before chitosan coating, aggregation of calcium phosphate was prevented and resulted in the formation of size-controlled composite materials.
Acknowledgments
This research was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIP) (No. 2011-0030121 and No. 2017R1A2B1012074).
References
- Mazaheri, M, Eslahi, N, Ordikhani, F, Tamjid, E, Simchi, A nanomedicine applications in orthopedic medicine: state of the art, Int J Nanomed, (2015), 10(1), p6039-54.
- Lee, MH, You, CK, Lee, HJ, Kwon, TY, Kim, KH, The effect of interconective micropores of biphasic calcium phosphate scaffold on osteoblast cellular behavior, Kor J Dent Mater, (2013), 40(4), p359-65.
-
Son, SK, Choi, IT, Ko, YM, Evaluation of calcium phosphate film coated dental implant with various electrochemical parameter, Kor J Dent Mater, (2017), 44(1), p43-51.
[https://doi.org/10.14815/kjdm.2017.44.1.043]

-
Tugulu, S, Lowe, K, Scharnweber, D, Schlottig, F, Preparation of superhydrophilic microrough titanium implant surfaces by alkali treatment, J Mater Sci Mater Med, (2010), 21, p2751-63.
[https://doi.org/10.1007/s10856-010-4138-x]

-
Jeon, WY, Han, AL, Surface modification of Ti-6Al-4V alloy by anodic oxidation and cyclic precalcification treatments, Kor J Dent Mater, (2016), 43(1), p1-8.
[https://doi.org/10.14815/kjdm.2016.43.1.1]

-
Ajami, E, Zinsou, KFA, Calcium phosphate growth at electropolished titanium surfaces, J Funct Biomater, (2012), 3, p327-48.
[https://doi.org/10.3390/jfb3020327]

-
Milleret, V, Tugulu, S, Schlottig, F, Hall, H, Alkali treatment of microrough titanium surfaces affects macrophage/monocyte adhesion, platelet activation and Architecture of blood clot formation, Eur Cells Mater, (2011), 21, p430-44.
[https://doi.org/10.22203/ecm.v021a32]

- Hsu, HC, Wu, SC, Hsu, SK, Chuang, SH, Ho, WF, Surface modification of commercially pure Ti treated with aqueous NaOH treatment and ethyl alcohol aging, J Med Biol Eng, (2012), 33(3), p331-6.
-
Wang, XX, Hayakawa, S, Tsuru, K, Osaka, A, A comparative study of in vitro apatite deposition on heat-, H2O2-, and NaOH-treated titanium surfaces, J Biomed Mater Res, (2001), 54, p172-8.
[https://doi.org/10.1002/1097-4636(200102)54:2<172::aid-jbm3>3.0.co;2-#]

-
Ahmed, S, Ikram, S, Chitosan based scaffolds and their applications in wound healing, Achiev Life Sci, (2016), 10(1), p27-37.
[https://doi.org/10.1016/j.als.2016.04.001]

-
Zhang, Y, Ni, M, Ratner, B, Calcium phosphate-chitosan composite scaffolds for bone tissue engineering, Tissue Eng, (2002), 9(2), p337-45.
[https://doi.org/10.1089/107632703764664800]

-
Aranaz, I, Campos, EM, Vicente, CM, Civantos, A, Arguelles, SG, Monte, F, Macroporous calcium phosphate/chitosan composites prepared via unidirectional ice segregation and subsequent freeze-drying, Materials, (2017), 10(5), p516.
[https://doi.org/10.3390/ma10050516]

-
Pighinelli, L, Kucharska, M, Properties of microcrystalline chitosan-calcium Phosphate complex composite, J Biomater Nanobiotechnol, (2013), 4, p20-9.
[https://doi.org/10.4236/jbnb.2013.44a003]

-
Lu, X, Wang, YB, Liu, YR, Wang, JX, Qu, SX, Feng, B, et al. , Preparation of HA/chitosan composite coatings on alkali treated titanium surfaces through sol–gel techniques, Mater Lett, (2007), 61, p3970-3.
[https://doi.org/10.1016/j.matlet.2006.12.089]

-
Schneider, M, Günter, C, Taubert, A, Co-deposition of a hydrogel/calcium phosphate hybrid layer on 3D printed poly(lactic acid) scaffolds via dip coating: towards automated biomaterials fabrication, Polymers, (2018), 10, p275.
[https://doi.org/10.3390/polym10030275]

-
Weng, J, Wang, M, Chen, J, Plasma-sprayed calcium phosphate particles with high bioactivity and their use in bioactive scaffolds, Biomaterials, (2002), 23, p2623-9.
[https://doi.org/10.1016/s0142-9612(01)00393-3]

-
Bosco, R, Beucken, JVD, Leeuwenburgh, S, Jansen, J, Surface engineering for bone implants: a trend from passive to active surfaces, Coatings, (2012), 2, p95-119.
[https://doi.org/10.3390/coatings2030095]

-
Aparicio, C, Padrós, A, Gil, FJ, In vivo evaluation of micro-rough and bioactive titanium dental implants using histometry and pull-out tests, J Mech Behav Biomed, (2011), 4(8), p1672-82.
[https://doi.org/10.1016/j.jmbbm.2011.05.005]

-
Kato, H, Nakamura, T, Nishiguchi, S, Matsusue, Y, Kokubo, T, Bonding of alkali-and heat-treated tantalum implants to bone, J Biomed Mater Res, (2000), 53(1), p28-35.
[https://doi.org/10.1002/(sici)1097-4636(2000)53:1<28::aid-jbm4>3.0.co;2-f]

-
Wang, YQ, Pan, L, Tao, J, Wang, T, Bioactive porous titania formed by two‐step chemical treatment of titanium substrates under high intensity ultrasonic field, Surf Eng, (2011), 27(1), p46-50.
[https://doi.org/10.1179/174329409x455359]

-
Song, HJ, Park, YJ, Moon, HJ, Luong, LN, Kohn, DH, Effects of Protein-Simulated Body Fluid Mixing Methods on Characteristics of Bone-Like Mineral, Mater Sci Eng C Mater Biol Appl, (2012), 32(8), p2501-7.
[https://doi.org/10.1016/j.msec.2012.07.032]

-
Kim, HM, Kokubo, T, Fujibayashi, S, Nishiguchi, S, Nakamura, T, Bioactive macroporous titanium surface layer on titanium substrate, J Biomed Mater Res, (2000), 52(3), p553-7.
[https://doi.org/10.1002/1097-4636(20001205)52:3<553::aid-jbm14>3.0.co;2-x]

-
Teixeira, RLP, Godoy, GCD, Pereira, MM, Calcium Phosphate Formation on Alkali-Treated Titanium Alloy and Stainless Steel, Materials Research, (2004), 7(2), p299- 303.
[https://doi.org/10.1590/s1516-14392004000200013]

-
Cacciotti, I, Bianco, A, Lombardi, M, Montanaro, L, Mg-substituted hydroxyapatite nanopowders: synthesis, thermal stability and sintering behavior, J Eur Ceram Soc, (2009), 29(14), p2969-78.
[https://doi.org/10.1016/j.jeurceramsoc.2009.04.038]

-
Finisie, MR, Josué, A, Fávere, VT, Laranjeira, MCM, Synthesis of calcium-phosphate and chitosan bioceramics for bone regeneration, An Acad Bras Cienc, (2001), 73(4), p525-32.
[https://doi.org/10.1590/s0001-37652001000400006]

- Kim, HE, Himeno, T, Kawashita, M, Kokubo, T, Nakamura, T, The mechanism of biomineralization of bone-like apatite on synthetic hydroxyapatite: An in vitro assessment, J R Soc Interface, (2004), 1, p17-22.
-
Kim, HM, Miyaji, F, Kokubo, T, Nishiguchi, S, Nakamura, T, Graded surface structure of bioactive titanium prepared by chemical treatment, J Biomed Mater Res, (1999), 45, p100-7.
[https://doi.org/10.1002/(sici)1097-4636(199905)45:2<100::aid-jbm4>3.3.co;2-s]

-
Kim, HM, Miyaji, F, Kokubo, T, Nakamura, T, Preparation of bioactive Ti and its alloys via simple chemical surface treatment, J Biomed Mater Res, (1996), 32, p409-17.
[https://doi.org/10.1002/(sici)1097-4636(199611)32:3<409::aid-jbm14>3.0.co;2-b]