
The translucent and color change of nanofilled composite resin according to resin shades and staining solutions
This study examined the color changes in a resin composite with different shades after exposure to different drink media. A nanohybrid resin composite (Filtek Z350 XT) was exposed to a pH 6 solution for 2 weeks and then immersed in a staining solution (Coffee or Grape juice) or distilled water (as control) for 3 days. The color coordinates of the specimen before and after immersion in the staining solutions were measured using a spectrophotometer. The color difference (ΔE ) and the change in the translucency parameter (ΔTP ) were calculated using the CIEL*a*b* system. The data was statistically analyzed using ANOVA and a Tukey’s test. The ΔE of the specimens tested differed significantly according to the staining solutions (p<0.05) and the shades of the composite resins (p<0.05). Coffee and B1 shade showed the greatest color change. The values of TP after immersion in the staining beverages decreased except for distilled water. The absolute ΔTP values of grape juice were higher than the others regardless of the shades. Overall, the color stability of the resin composites differs according to the staining media used and shade of the composite resins.
Keywords:
Color stability, composite resin, staining solution, translucency parameter, resin shadeINTRODUCTION
Dental resin composite is the most common esthetic restorative material. Shade matching of the resin composite restoration is of critical concern in esthetic dentistry. The color of the materials should remain stable over a long period in the oral environment. On the other hand, resin composites are susceptible to various degrees of discoloration upon prolonged exposure to the oral environment (Uchida et al, 1998; Bagheri et al, 2005; Correr et al, 2012). The discoloration of composites often results in the need to replace the restoration, which is expensive and time consuming (Millar et al, 1997; van Dijken, 2001).
The discoloration of resin composites can be induced internally or externally. Intrinsic factors involve discoloration of the resin material itself, such as a change in the resin matrix, the interface of the matrix and the fillers (Sarafianou et al, 2007; Barutcigil and Yildiz, 2012) The extrinsic factors for discoloration include staining by the adsorption or absorption of colorants due to contamination from exogenous sources (Asmussen and Hansen, 1986; Bagheri et al, 2005).
In the oral cavity, tooth-colored restorative dental materials are exposed continuously to saliva and water in the oral environment. In particular, the restored composite resins always interact with water. Although composite resin interacts with extraneous water, the water uptake into the composite resin occurs through a diffusion-controlled process (Söderholm et al, 1984). Therefore, water sorption is a diffusion-controlled process in the resin matrix, which can lead to degradation and debonding of the filler matrix interlayer (Braden, 1984; Söderholm et al, 1984). This can deteriorate the mechanical and optical properties (Oysaed and Ruyter, 1986). The color of resin composite materials is affected indirectly by degradation with time due to water uptake, and the subsequent hydrolysis and chemical reactions (Larsen and Munksgaard, 1991).
Although there have been many studies on the color stability of resin composites (Guler et al, 2005; Fujita et al, 2006; Mohan et al, 2008; Ardu et al, 2010), these studies focused mainly on the influence of tea, coffee and soft drinks on the color stability of resin composites immediately after polymerization. Information on the difference in the color stability according to the staining agents after the hydrolysis of resin composites by water is limited.
This study examined the effects of staining solutions on the color stability of restorative resin composites after degradation by water uptake. The color stability was tested by examining the color difference (ΔE) and difference in the translucency parameter (TP) of one resin composite with four different shades in the three solutions. The null hypothesis tested in this study was that the staining solutions have no effect on the color change and TP difference of the resin composite with different shades after degradation by water uptake.
MATERIALS AND METHODS
For the study, the nano-filled composite resin (Filtek Z350 XT, 3M ESPE, St. Paul, MN, USA) with four different shades (A2, A3, B1 and B2) was used. A LED light-curing unit (L.E.Demetron, Kerr, Danbury, CT, USA) was used for light curing. To prepare the specimens, a metal ring mold (inner diameter: 8mm; thickness: 2 mm; n=10 for each condition) was filled with resin and light cured for 40 seconds with a light intensity of 1000 mW/cm2. The light cured specimens were then removed from the mold and aged for 24 hours in a dark dry chamber at 37 °C. The aged specimens were then immersed in the solutions at pH 6 for 14 days. Distilled water was mixed with dilute acetic acid to make the pH 6 solutions. All processes were carried out at 23±1°C and 60±3% humidity.
After immersion in the pH 6 solutions, each specimen was tested by immersion in one of the following three test solutions for 3 days: distilled water (DW); coffee (CF) - 0.45g of instant coffee (Taster’s Choice, Nestlé USA, Inc., Glendale, CA, USA) powder/50cc distilled water, without sugar or cream; or grape juice (GJ) (Fresh grape juice, Seoul Diary Co., Seoul, Korea).
A spectrophotometer (CM-3600d, Konica Minolta, Osaka, Japan) was used to measure the color of the specimens relative to the standard illuminations D65. The initial color of the light-cured specimens was measured by placing the specimen at the center of the target mask in reflectance (%R) mode with white and black backgrounds. This target mask had a 6 mm hole at the center. This hole enables consistency in specimen placement during the measurements. After the first color measurement, each specimen was immersed in the designated test solution for 3 days. During immersion, the solutions were replaced daily. After 1 and 3 days, the specimens were removed from the immersion solution and rinsed in running water. After removing the water remaining on the surface with tissue paper, the specimen was placed into the hole at the center of the target mask and its reflectance measured again. From the measured reflectance data, the CIEL*a*b* color values were evaluated using the internal software of the measurement system. The CIEL*a*b* color system is a three-dimensional color measurement system. Here, L* refers to the lightness coordinate, which has a value ranging from 0 for perfect black to 100 for perfect white, and a* and b* are the chromaticity coordinates on the green–red (-a*=green; +a*=red) and blue–yellow (-b*=blue; +b*=yellow) axes, respectively. ΔE was obtained using the following formula:
ΔE= [ (ΔL* )2+(Δa* )2+(Δb* )2]1/2,
where Δ is the difference between the first and second measurements.
The TP values were determined by calculating the color difference between the readings over a black and white background for the same specimen using the following formula:
TP = [(L*B-L*W)2+(a*B-a*W)2+(b*B-b*W)2]1/2,
where subscripts ‘B’ and ‘W’ refer to the color coordinates over a black and white background, respectively. The change in the translucency parameter, ΔTP (TP after immersion in the test solutions minus TP before immersion in the test solutions), was calculated for each specimen.
The results of the color change according to the test solutions and shades of the composite resin were analyzed by two-way ANOVA. A post hoc Tukey test was then performed using a multiple comparison procedure. A p value <0.05 was considered significant.
RESULTS
Tables 1 and 2 list the CIEL*a*b* color coordinates and color changes in the specimens before and after immersion in the test solutions. The specimens of the A2 and A3 shades had lower L*and higher b*values than those of the B1 and B2 shades. The range of ΔL*, Δa*and Δb* values were -0.35 ~ -4.57, -0.02 ~ 0.94 and -0.30 ~7.93, respectively. The specimens immersed in grape juice showed slightly higher ΔL*and Δa* values, whereas the specimens immersed in coffee showed higher Δb*values than the other solutions. The specimens of shade B1 showed higher Δb*values than the other shades.
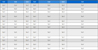
Mean and standard deviation values of color coordinates(ΔL*, Δa*, Δb*) after immersion in test solutions for 3 days
Table 3 lists the mean and standard deviations of the ΔE values of the resin composite with different shades after exposure to the three solutions. The ΔE of the tested specimens differed significantly among the three staining solutions (p<0.05). The specimens immersed in coffee showed a significantly greater color differences than those of the other cases (p<0.05). For the shades of the resin composite, there was significant differences between the shades, and the specimens of shade B1 showed larger color differences than the others.

Color changes (ΔE*) and standard deviations in the specimens of various shades after 3 days immersion of test solution
Table 4 lists the TP values and TP difference (ΔTP) of the specimens before and after immersion in the staining solutions for 3 days. Higher ΔTP values were observed in the specimens of immersed in grape juice regardless of the shade.
DISCUSSION
Color stability is very important in recent restorative composite resins because of the emphasis on esthetics. Generally, the color change might be related easily to the nature of the resin matrix. Similarly, the filler content appears to play an important role in the composite color stability (Rodrigues et al 2008a; Rodrigues et al, 2008b) On the other hand, there is little evidence to confirm whether the degree of degradation or water uptake prior to immersion in the staining solutions affects the color in the composite resin according to the shade. This study was carried out to determine if the water uptake at special periods, e.g. before immersion in the staining solution, causes changes in the composite color.
The beverages tested in this study induced varying degrees of discoloration in the resin composites tested. Coffee caused the greatest discoloration (ΔE=3.56-8.96)in all shades, followed by grape juice (ΔE=3.29-4.80). The color difference value, ΔE, is expressed as the relative color change between the repeated color measurements. In this system, the ΔE value of 3.3 is considered clinically perceptible and therefore unacceptable (Schulze et al, 2003a). In the present study, the color change values for all shades of resin composites in coffee and grape juice were greater than or equal to 3.3. These values were considered visually perceptible and clinically unacceptable. On the other hand, only a slight color change (ΔE=0.49-0.64) was observed in distilled water (control group), which was considered visually undetectable.
In this study, the composites in all staining solutions, especially coffee, showed visible discoloration. This is in line with the findings obtained in other studies (Um and Ruyter, 1991; Iazzetti et al, 2000; Lu et al, 2005; Villalta et al, 2006). The discoloration in coffee was attributed to adsorption and absorption. Coffee includes a yellow color in materials with low polarity that is released slowly and penetrates into the organic part of the materials. This might be due to the matching of the polymeric materials with the yellow color-causing materials in the coffee (Malekipour et al, 2012).
In the present study, the specimens immersed in grape juice showed slight higher ΔL* and Δa* values, and the specimens immersed in coffee showed higher Δb* values than in the other solutions. The change in the b* chromaticity coordinate (the yellowness) appeared to be the major color change in the specimens immersed in coffee because the changes in the other coordinates (i.e., L*and a*) were smaller (exhibiting more scatter) than the changes in the b*coordinate values in the specimens immersed in coffee. This is also evident from the fact that the changes in the E values occurred in a similar manner to that of the Δb* changes as a function of the shade changes, indicating that the total color change was a strong function of the changes occurring in the b*chromaticity coordinate. Therefore, it appears that the major changes observed in the CIEL*a*b* occurred in the b* coordinate. This increase in the b* coordinate reflects a shift to a more yellow color range.1)
Previous studies (Patel et al, 2004; Bagheri et al, 2005; Ertas et al, 2006) reported that the greatest color change was observed when the materials were immersed in red wine. On the other hand, they did not explain whether staining was caused by the alcohol or by the presence of pigments in the wine. In order to eliminate these variations, grape juice as an alternative beverage was used in the present study.
In this study, the effects of the shades on the color stability of composites were statistically significant. A significant difference was observed between the shades of resin composite, and the specimens of shade B1 showed the largest color differences, followed by B2, A2 and A3. These results show that the main effect of the shade variation is a general tendency for decreased color stability with increasing lightness of shade. A previous study (Brauer, 1988) performed a semi-quantitative comparison and reported that lighter shades showed more visible color changes than darker shades. Hosoya (Hosoya, 1999) and Schulze et al. (Schulze et al, 2003b) also reported that lighter or less chromatic shades tended to show larger color changes than more chromatic or darker shades.
According to Uchida et al. (Uchida et al, 1998), the greater discoloration associated with the lighter shades might result from one of two factors: (a) discoloration through environmental breakdown of the polymer leading to the release of monomers and a shift of color from the cured resin to that of the monomers, and (b) the environmental effects on the retention and/ or stability of pigments and other additives in polymer formulations.
Regarding the TP in the present study, TP is the color difference between a uniform thickness of the material on a black and white background, and corresponds directly to common visual assessments of translucency. In this study, the values of TP after immersion in the staining beverages decreased with the exception of the DW group. Higher ΔTP values were observed in the specimens immersed in grape juice regardless of the shade. The darkening of the composite resin immersed in the grape juice group might also affect the TP value. In summary, the discoloration of a composite resin is affected by a number of factors, such as the chemical reactivity, exposure to various energy sources, water sorption, diet, and surface smoothness of the restoration (Um and Ruyter, 1991; Lang et al, 1998; Koishi et al 2001; Sen et al, 2002; Eliades et al, 2004). Among them, the susceptibility to a color change in composite resins is most probably related to the degree of water sorption and the hydrophilicity of the resin matrix. The staining solutions affected the color stability of the restorative resin composites after degradation by water uptake. Despite the short staining period of the present study (3 days), coffee and grape juice seriously stained the tested materials. Therefore, the null hypothesis tested in this study, i.e., the staining solutions have no effect on the color change and the TP difference in the resin composite with different shades after the degradation by water uptake, was rejected. In clinical aspects, these results provide information on the color stability of resin composites and the staining potential of some drinks commonly consumed in daily life.
CONCLUSIONS
Within the limitation of this study, ΔE of the tested specimens differed significantly according to the staining solutions (p<0.05) and the shades of the composite resins (p<0.05). Coffee and B1 shade exhibited the greatest color change. The color change in these staining agents was visually perceptible and clinically unacceptable. The values of TP after immersion of the staining beverages decreased except for distilled water, and the absolute ΔTP values of grape juice were higher than the others regardless of the shades. In conclusion, the color stability of resin composites differs according to the staining media and shade of the composite resins. In clinical practice, patients should be aware of the staining effects of the drinks tested in this study, and practitioners should consider the staining susceptibility of the resin composites.
Acknowledgments
This study was supported by 2014 Clinical Research Grant, Pusan National University Dental Hospital
References
- S Ardu, V Braut, D Gutemberg, I Krejci, D Dietschi, AJ Feilzer, A long-term laboratory test on staining susceptibility of esthetic composite resin materials, Quintessence Int, (2010), 41, p695-702.
-
E Asmussen, EK Hansen, 1986). Surface discoloration of restorative resins in relation to surface softening and oral hygiene, Scand J Dent Res, (1986), 94, p174-177.
[https://doi.org/10.1111/j.1600-0722.1986.tb01382.x]

-
R Bagheri, MF Burrow, M Tyas, Influence of food simulating solutions and surface finish on susceptibility to staining of aesthetic restorative materials, J Dent, (2005), 33, p389-398.
[https://doi.org/10.1016/j.jdent.2004.10.018]

-
C Barutcigil Yıldız M, Intrinsic and extrinsic discolouration of dimethacrylate and silorane based composites, J Dent, (2012), 40(Suppl. 1), pe57-e63.
[https://doi.org/10.1016/j.jdent.2011.12.017]

- M Braden, Water absorption characteristics of dental microfine composite filling materials.II Experimental materials, Biomaterials, (1984), 5, p373-375.
-
GM Brauer, Color changes of composites on exposure to various energy sources, Dent Mater, (1988), 4, p55.
[https://doi.org/10.1016/S0109-5641(88)80090-3]

- GM Correr, RC Bruschi Alonso, F Baratto-Filho, L Correr-Sobrinho, MA Sinhoreti, RM Puppin-Rontani, In vitro long-term degradation of aesthetic restorative materials in food-simulating media, Acta Odontol Scand, (2012), 70, p101-108.
- T Eliades, C Gioka, M Heim, G Eliades, M Makou, Color stability of orthodontic adhesive resins, Angle Orthod, (2004), 74, p391-393.
-
E Ertas, AU Guler, AC Yucel, H Koprulu, E Guler, Color stability of resin composites after immersion in different drinks, Dent Mater J, (2006), 25, p371-376.
[https://doi.org/10.4012/dmj.25.371]

-
M Fujita, S Kawakami, M Noda, H Sano, 2006). Color change of newly developed esthetic restorative material immersed in food-simulating solutions, Dent Mater J, (2006), 25, p352-359.
[https://doi.org/10.4012/dmj.25.352]

-
AU Guler, F Yilmaz, T Kulunk, E Guler, S Kurt, Effects of different drinks on stainability of resin composite provisional restorative materials, J Prosthet Dent, (2005), 94, p118-124.
[https://doi.org/10.1016/j.prosdent.2005.05.004]

- Y Hosoya, Five-year color changes of lightcured resin composites: influence of light-curing times, Dent Mater, (1999), 15, p268-274.
- G Iazzetti, JO Burgess, D Gardiner, A Ripps, Color stability of fluoride-containing restorative materials, Oper Dent, (2000), 25, p520-525.
- Y Koishi, N Tanoue, H Matsumura, M Atsuta, Color reproducibility of a photo-activated prosthetic composite with different thicknesses, J Oral Rehabil, (2001), 28, p799-804.
- R Lang, M Rosentritt, A Leibrock, M Behr, G Handel, Color stability of provisional crown and bridge restoration materials, Br Dent J, (1998), 185, p468-471.
-
IB Larsen, EC Munksgaard, Effect of human saliva on surface degradation of composite resins, Scand J Dent Res, (1991), 99, p254-261.
[https://doi.org/10.1111/j.1600-0722.1991.tb01893.x]

-
H Lu, LB Roeder, L Lei, JM Powers, Effect of surface roughness on stain resistance of dental resin composites, J Esthet Restor Dent, (2005), 17, p102-108.
[https://doi.org/10.1111/j.1708-8240.2005.tb00094.x]

- MR Malekipour, A Sharafi, S Kazemi, S Khazaei, F Shirani, Comparison of color stability of a composite resin in different color media, Dent Res J (Isfahan), (2012), 9, p441-446.
-
BJ Millar, PB Robinson, AT Inglis, Clinical evaluation of an anterior hybrid composite resin over 8 years, Br Dent J, (1997), 182, p26-30.
[https://doi.org/10.1038/sj.bdj.4809289]

- M Mohan, Z Shey, J Vaidyanathan, TK Vaidyanathan, S Munisamy, M Janal, Color changes of restorative materials exposed in vitro to cola beverage, Pediatr Dent, (2008), 30, p309-316.
- H Oysaed, IE Ruyter, Composites for use in posterior teeth. Mechanical properties tested under dry and wet conditions, J Biomed Mater Res, (1986), 20, p261-271.
-
SB Patel, VV Gordan, AA Barrett, C Shen, The effect of surface finishing and storage solutions on the color stability of resin-based composites, J Am Dent Assoc, (2004), 135, p587-594.
[https://doi.org/10.14219/jada.archive.2004.0246]

- SA Rodrigues Jr, JL Ferracane, A Della Bona, Flexural strength and Weibull analysis of a microhybrid and a nanofill composite evaluated by 3- and 4-point bending tests, Dent Mater, (2008a), 24, p426-431.
-
SA Rodrigues Jr, SS Scherrer, JL Ferracane, A Della Bona, Microstructural characterization and fracture behavior of a microhybrid and a nanofill composite, Dent Mater, (2008b), 24, p1281-1288.
[https://doi.org/10.1016/j.dental.2008.02.006]

-
A Sarafianou, S Iosifidou, T Papadopoulos, G Eliades, Color stability and degree of cure of direct composite restoratives after accelerated aging, Oper Dent, (2007), 32, p406-411.
[https://doi.org/10.2341/06-127]

- KA Schulze, SJ Marshall, SA Gansky, GW Marshall, Color stability and hardness in dental composites after accelerated aging, Dent Mater, (2003a), 19, p612-619.
- KA Schulze, J Tinschert, SJ Marshall, GW Marshall, Spectroscopic analysis of polymer-ceramic dental composites after accelerated aging, Int J Prosthodont, (2003b), 16, p355-361.
- D Sen, G Goller, H Issever, The effect of two polishing pastes on the surface roughness of bis-acryl composite and methacrylate-based resins, J Prosthet Dent, (2002), 88, p527-532.
- KJ Söderholm, M Zigan, M Ragan, W Fischlschweiger, M Bergman, Hydrolytic degradation of dental composites, J Dent Res, (1984), 63, p1248-1254.
-
H Uchida, J Vaidyanathan, T Viswanadhan, TK Vaidyanathan, Color stability of dental composites as a function of shade, J Prosthet Dent, (1998), 79, p372-377.
[https://doi.org/10.1016/S0022-3913(98)70147-7]

- CM Um, IE Ruyter, Staining of resin-based veneering materials with coffee and tea, Quintessence Int, (1991), 22, p377-386.
- JW van Dijken, Durability of new restorative materials in Class III cavities, J Adhes Dent, (2001), 3, p65-70.
-
P Villalta, H Lu, Z Okte, F Garcia-Godoy, JM Powers, Effects of staining and bleaching on color change of dental composite resins, resins. J Prosthet Dent, (2006), 95, p137-142.
[https://doi.org/10.1016/j.prosdent.2005.11.019]
