Compositional tuning of gentamicin-loaded alginate-chitosan beads for optimized drug release and biological properties
Abstract
This study investigated the encapsulation of gentamicin sulfate (Gn) into beads composed of sodium alginate and chitosan (Ch) conjugates using ionotropic gelation, focusing on how compositional variations in chitosan (0.1–1.0 wt%) and gentamicin (0.003–0.3 wt%) affect their physicochemical and biological performance. Scanning electron microscopy revealed composition-dependent changes in surface morphology that directly affected drug release behavior. Comprehensive characterization using FT-IR and TGA confirmed successful Gn incorporation and the thermal stability of the composite beads. Swelling behavior was strongly influenced by both polymer and drug content: higher Ch concentrations enhanced water uptake through increased hydrophilicity, whereas greater Gn loading reduced swelling by forming denser ionic crosslinks within the polymer network. Adjusting the Ch and Gn concentrations could control the Gn release rate from Gn-loaded alginate-chitosan beads in phosphate-buffered saline, enabling formulation-dependent tuning of both release rate and duration. All Gn-loaded beads prepared in this study demonstrated antimicrobial activity against Staphylococcus aureus. In vitro studies showed that beads loaded with varying Gn concentrations (0.003% to 0.3%) maintained >70% cell viability in human gingival fibroblasts (HGFs). These findings highlight the potential of compositionally optimized alginate-chitosan beads as biocompatible platforms for localized antimicrobial drug delivery.
초록
본 연구에서는 이온겔화법을 이용하여 제조된 겐타마이신 황산염(Gn) 함유 알긴산-키토산 복합 비드의 조성 변화가 물리화학적 및 생물학적 특성에 미치는 영향을 평가하였다. 키토산(0.1–1.0 wt%) 및 겐타마이신(0.003–0.3 wt%)의 함량을 조절하여 제조한 비드의 표면 형태는 주사전자현미경(SEM)을 통해 관찰되었으며, 조성에 따른 표면 형태적 차이가 확인되었고, 이러한 표면 형태의 차이는 약물 방출 거동과 밀접한 연관성을 나타냈다. Fourier 변환 적외선 분광법(FT-IR) 및 열중량 분석(TGA)을 통해 겐타마이신이 비드 내에 성공적으로 함입되었으며, 복합체의 열적 안정성도 확보되었음을 확인하였다. 팽윤 특성은 고분자 조성과 약물 함량에 따라 크게 달라졌으며, 키토산 농도가 증가할수록 친수성이 증가하여 수분 흡수가 증가하였고, 반대로 겐타마이신 농도가 증가할수록 고분자 네트워크 내의 이온성 가교 결합이 조밀하게 형성되어 팽윤이 감소되었다. 키토산과 겐타마이신의 조성 조절을 통해 인산완충생리식염수 내에서 약물 방출 속도와 지속 시간을 정밀하게 제어할 수 있었다. 모든 조성의 Gn-함유 비드는 황색포도상구균(Staphylococcus aureus)에 대해 항균 활성을 나타냈으며, human gingival fibroblasts를 이용한 세포적합성 평가에서도 70% 이상의 세포 생존율을 보여 양호한 생체적합성을 입증하였다. 이러한 결과는 조성 제어가 가능한 알긴산-키토산 비드가 국소 항균 약물 전달 플랫폼으로서의 높은 응용 가능성을 지님을 시사한다.
Keywords:
Chitosan, Alginate, Gentamicin, Controlled release, Antimicrobial activity키워드:
키토산, 알지네이트, 겐타마이신, 제어방출, 항균활성Introduction
Many drug delivery systems employ naturally derived biopolymers with biodegradability, biocompatibility, and controlled drug release properties (1, 2). Among the various delivery formats, bead carriers prepared from a combination of two or more different polymers perform better than carriers using a single polymer. Beads derived from natural sources, such as polysaccharides, are particularly attractive for localized and sustained drug delivery because of their excellent biocompatibility, biodegradability, high water absorption, and non- cytotoxicity compared with beads synthesized artificially (3).
Alginate is a negatively charged polysaccharide linear copolymer comprising varying proportions of β-D-mannuronic acid and α-L-guluronic acid connected through β-1, 4-glycosidic bonds (4). Alginate is extracted from brown seaweeds and is renowned for its suitability in bead formation with diverse materials. It has extensive applications in wound care, controlled drug delivery, and as an additive in pharmaceuticals, food products, and the cosmetic industry. The swelling characteristics of alginate composite hydrogels represent optimal superabsorbent quality for various solutions (5). In a one-step manufacturing process, droplets of alginate solution are introduced into an aqueous solution containing a gelling agent for alginate (such as calcium ions) and chitosan. When ionically cross-linked with positively charged polymers such as chitosan, alginate forms stable, biocompatible matrices suitable for encapsulating a variety of therapeutic agents
Chitosan, a biopolymer derived by N-deacetylating the polysaccharide chitin, has gained significance in the pharmaceutical industry due to its distinctive cationic nature, biocompatibility, non-toxicity, and biodegrada-bility, making it a functional copolymerizing compound for the controlled release of water-soluble medicaments (6).
Gentamicin sulfate (Gn), a broad-spectrum aminoglycoside antibiotic, is widely used for treating infections associated with orthopedic implants and surgical procedures. However, its systemic administration often associated with dose-dependent toxicity and suboptimal local concentrations, underscoring the need for more effective localized delivery systems. Gn can form gels through ionic interactions with the positively charged amino groups of chitosan. Calcium chloride (CaCl2), acting as a physical cross-linker, rapidly binds and cross-links with an α-L-guluronic acid moieties of sodium alginate via ion exchange facilitated by Ca2+ cations, forming an egg-box structured gel that enhances matrix stability. This gel network is suitable for applications in biomedicine, particularly as a substrate for platelet and cell attachment (7, 8). Recently, biopolymer complexes have gained attention for designing controlled drug delivery systems (DDS). Cationic chitosan creates gels through ionic cross-linking with anionic counterions of sodium alginate. Active components released from drug-releasing biopolymers traverse various biological barriers to reach the intended target area. This DDS enhances drug effectiveness by allowing controlled release, which decreases dosing frequency and reduces potential side effects. Typically, the DDS uses polymeric carriers, which possess functional groups, to facilitate the binding of drug molecules (9).
Although prior studies have demonstrated the feasibility of incorporating Gn into alginate-chitosan matrices, most were limited to single-point formulations and did not comprehensively evaluate the impact of varying composition on the structural, physicochemical, and biological properties of the beads (9). A comprehensive analysis of how chitosan and gentamicin concentrations influence bead performance is needed to guide the design of optimized drug carriers.
In this study, we systematically investigated the effect of varying chitosan and gentamicin content on the morphology, swelling behavior, drug release kinetics, antibacterial activity, and cytocompatibility of Gn-loaded alginate-chitosan beads. This research not only advances the fundamental understanding of composition–property relationships in biopolymer-based drug carriers but also supports their potential application as localized antimicrobial delivery platforms for oral and maxillofacial applications.
Materials and Methods
1. Materials
Sodium alginate, calcium chloride dihydrate (CaCl2·2H2O), and gentamicin sulfate were obtained from Sigma-Aldrich (Korea). Chitosan (85% deacetylation, molecular weight 103 kDa) and gentamicin solution (10 mg/mL in deionized water, liquid, 0.1 µm filtered) were also purchased from Sigma-Aldrich (Korea). All reagents were of analytical grade and used without further purification.
2. Fabrication of gentamicin-loaded chitosan-alginate beads
A 2% (w/v) aqueous solution of sodium alginate (viscosity 300 cp) was prepared at 25℃. A 3% chitosan solution (w/v) was prepared by dissolving 3 g of chitosan in 100 mL of 1% (v/v) acetic acid solution at room temperature. The sodium alginate solution was used as a dope in the gelation process for preparing the chitosan-alginate beads. Homogeneous aqueous mixtures of chitosan (Ch) and CaCl2·2H2O in varying proportions were used as coagulation solutions. Gn-loaded alginate-chitosan beads were prepared by dropping 100 mL alginate solution into a 200 mL CaCl2·2H2O solution containing 0.1–1.0% (w/v) of 3% Ch solution and 0.003–0.3% (w/v) of Gn under stirring for 15 min. The pH of the coagulation solution was adjusted to 5.5±0.1. A 20 mL of the alginate solution (dope) was dispensed using a 21-gauge needle into 50 mL of the coagulation solution under constant stirring at 200 rpm. The beads were maintained in the coagulation bath for 1 h to ensure complete cross-linking, then collected, thoroughly washed with distilled water, and freeze-dried at ‒40℃ for 24 h.
3. Characterization of beads
The surface morphology of the beads was examined using a field-emission scanning electron microscope (FE-SEM; S-4700, Hitachi, Tokyo, Japan). Fourier-transform infrared (FTIR) spectra of the beads were recorded using an FTIR spectrometer equipped with an attenuated total reflection (ATR) module (Tensor I, Bruker, Billerica, USA) in the 500–4000 cm-1 range with a resolution of 1 cm-1. Thermal stability of sodium alginate, Gn, and alginate-Ch-Gn beads (xCh-yGn; x = 0.1–1.0 wt%, y = 0.003–0.3 wt%) was evaluated by thermogravimetric analysis (TGA; Mettler Toledo, Greifensee, Switzerland). TGA was performed from 30℃ to 800℃ at a heating rate of 5 ℃/min under nitrogen atmosphere with a flow rate of 40 mL/min.
4. Swelling behavior
Swelling of the prepared beads was assessed in phosphate-buffered saline (PBS) with a pH of 7.4. Freeze-dried beads were immersed in PBS and removed periodically for weighing. Excess surface liquid was removed using filter paper prior to measurement. The swelling ratio (SR) was calculated using following equation (10):
where W0 and Wt denote the initial dry weight and the swollen weight of the beads at time t, respectively.
5. In vitro drug release
The release of Gn was evaluated for 24 h in PBS (pH 7.4). Dried beads (0.1 g) were immersed in 10 mL of PBS and maintained under constant agitation at 37℃ in a bio-oven. At predetermined intervals (0.5, 1, 3, 6, 9, 12, 24 h), 1 mL of the release medium was withdrawn for analysis, and an equivalent volume of fresh PBS was added to maintain constant total volume. The collected leachate solution was filtered through a 0.22 µm cellulose acetate filter, and Gn concentration was determined using an ACQUITY ultra-performance liquid chromatography (UPLC) H-Class system (Waters, Manchester, UK), coupled with a quadrupole time-of-flight tandem mass spectrometer (XEVO-G2XS QTof system, Waters, Manchester, UK).
6. Antibacterial activity of extracted gentamicin from beads
Dried beads (0.1 g) were placed into 10 mL of PBS at 37℃ for 24 h in a shaking incubator. Gn released from the beads was quantified using the test tube serial dilution method (11). Staphylococcus aureus (S. aureus; ATCC29213, American Type Culture Collection) served as the model bacterium. Bacterial stocks were stored at −80℃ in tryptic soy broth containing 50% glycerol. S. aureus was cultured on a brain heart infusion (BHI) agar plate at 37℃ for 24 h in a 5% CO2 incubator. The strains were grown overnight in BHI media supplemented with 1% glucose. Then, bacteria were sub-cultured in broth medium until they reached the mid-exponential phase. This study used a microdilution broth method to determine the minimal inhibitory concentration (MIC) of the antimicrobial agent. The cultured bacteria were seeded in a 96-well plate at a concentration of 1×105 cells/mL, and 100 µL of the extracted solution was treated and incubated at 37℃ for 24 h under 5% CO2. The optical density (OD) was measured at 600 nm using a microplate spectrophotometer (Epoch, BioTek Instruments, Winooski, USA). Antibacterial efficacy was calculated using the following equation:
where Ac represents the absorbance of the bacteria suspension cultured without-Gn leachate (control), while As refers to the absorbance of the suspension cultured with-Gn leachate (samples).
7. Cytotoxicity evaluation of extracted gentamicin
Cytotoxicity was evaluated using primary human gingival fibroblasts (HGF) collected from four periodontally and systemically healthy donors (no medication or general disease) having no signs of periodontal attachment loss and no bleeding on probing. All participants were patients at the University of Pennsylvania School of Dental Medicine. The study was reviewed and approved by the Institutional Review Board of the University of Pennsylvania (protocol #825886). Written informed consent was obtained from all participants. HGFs were cultured in a CO2 incubator using Dulbecco’s Modified Eagle Medium (DMEM; GIBCO, USA) supplemented with 10% fetal bovine serum, 1% penicillin/streptomycin, and 5 mL L-glutamine solution (GIBCO). The cultured cells were seeded in a 96-well plate at a density of 5×104 cells/mL and incubated at 37℃ for 24 h in a humidified 5% CO2 atmosphere. Gn extracted to DMEM solutions (GnE) from the beads was filtered before testing using 0.22 µm polyethersulfone syringe filters (Millipore, Billerica, USA). After 24 h, 100 µL of GnE was added to each well, and the cells were further incubated for another 24 h. DMEM without GnE served as the control. Cell viability was assessed using the MTT assay (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; Amresco, United States), conducted in triplicate (n=3). Absorbance at 570 nm was measured using the microplate spectrophotometer (Epoch, BioTek Instruments, Winooski, USA).
8. Statistical analysis
A one-way ANOVA was performed using GraphPad Prism version 10 (GraphPad Software, La Jolla, USA); a p-value of <0.05 was considered statistically significant.
Results
1. Characterization of the beads
The SEM images of the freeze-dried alginate-chitosan beads fabricated with the compositional variations in alginate, chitosan, and gentamicin concentrations are shown in Figure 1. SEM images of alginate-chitosan beads and Gn-loaded alginate-chitosan beads revealed an irregular and porous surface structure with variable surface pore sizes. The beads exhibited a spherical shape with a rough surface. By altering the content of chitosan and Gn, the size and surface morphology of the beads were identified. An increase in chitosan content notably influenced the size, leading to larger alginate-chitosan beads (Figure 1a-c). The inclusion of chitosan affected pore distribution within the beads. In the case of Gn-loaded alginate-chitosan beads, increasing the chitosan content from 0.1 to 1.0% induced a transition from a larger to a smaller and denser wrinkled surface (from left to right, Figure 1d → e → f), while increasing the Gn content from 0.003% to 0.3% resulted in deeper wrinkles (from top to bottom, Figure 1d → g → j, Figure 1e→ h → k, Figure 1f → i → l).
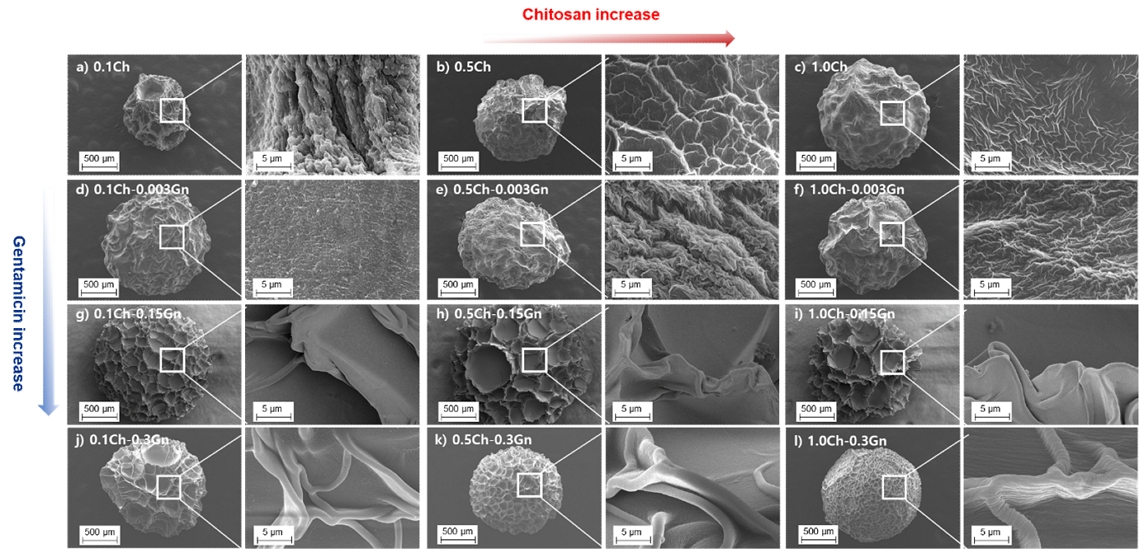
FE-SEM images of freeze-dried alginate-chitosan beads (xCh-yGn) with varying chitosan (Ch; 0.1%, 0.5%, 1.0%) and gentamicin (Gn; 0%, 0.003%, 0.15%, 0.3%) concentrations. Beads in (a–c) contain no Gn, whereas (d–f), (g–i), and (j–l) contain 0.003%, 0.15%, and 0.3% Gn, respectively. Chitosan content increases from left to right within each row. The right-side images show magnified views of the boxed regions of respective left-side images. Scale bars: 5 µm (left), 500 nm (right).
In Figure 2, the FT-IR spectra of various Gn-loaded alginate-chitosan beads are shown. From the spectra, a successful incorporation of Gn on the surface of alginate-chitosan beads could be identified. The broad absorption peak at 3250 cm–1 originated from the stretching vibration of the hydroxyl group in Gn-loaded alginate-chitosan beads and the overlapping stretching of N–H groups in chitosan and Gn. The asymmetric and symmetric vibrations of COO– of alginate appeared near 2932 cm–1. The characteristic absorption peaks of chitosan and alginate were observed at 1586, 1413, 1079, and 1029 cm–1(12). Reports show that the intense band at 1586 cm–1 and 1413 cm–1 are attributed to asymmetric and symmetrical C=O stretching. The FT-IR spectra of Gn-loaded alginate-chitosan beads confirmed the presence of Gn within the bead structure by the observation of characteristic Gn absorption bands at 1152 cm–1 and 884 cm–1, attributing to the C-O-C stretching, and at 1586 cm–1 attributing to the N-H vibration of Gn (13).
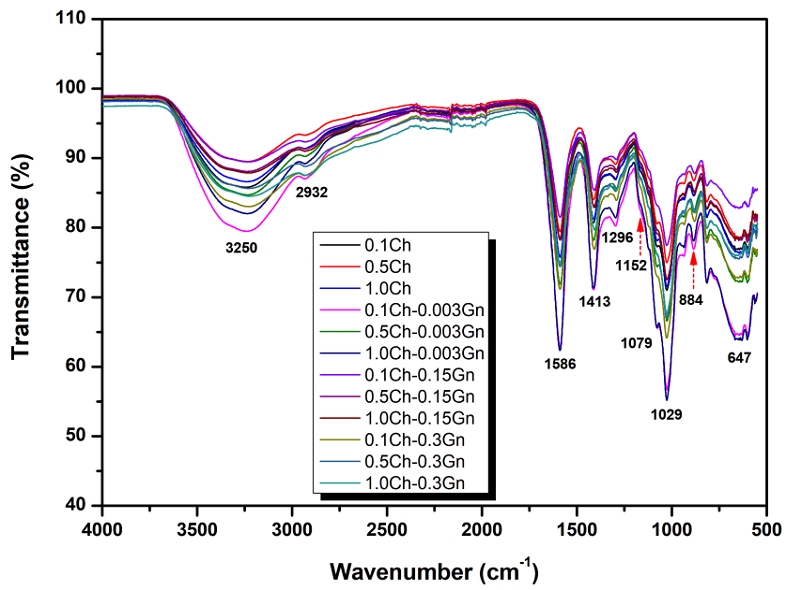
FT-IR spectra of gentamicin-loaded alginate-chitosan beads (xCh-yGn; x = 0.1, 0.5, 1.0% and y = 0.003, 0.15, 0.3%) with varying chitosan (x) and gentamicin (y) concentrations.
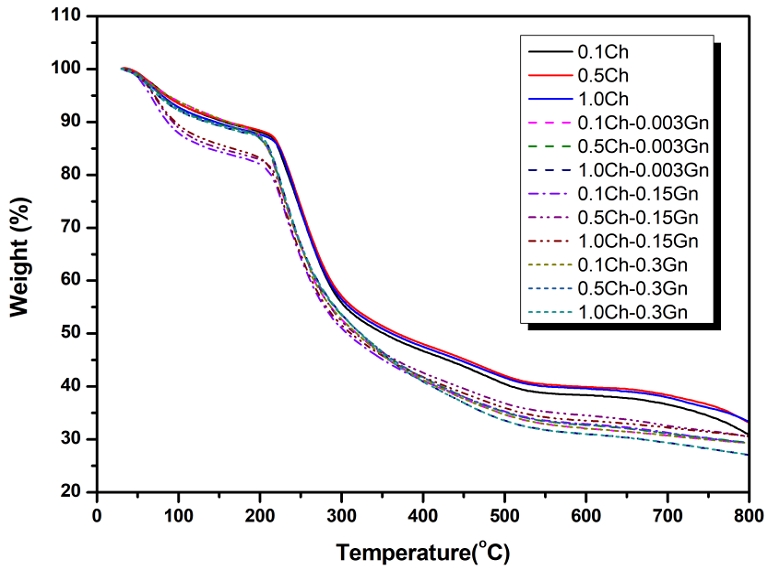
The TGA thermograms of alginate-chitosan beads and Gn-loaded alginate-chitosan beads are shown in Figure 3. The TGA profile demonstrated weight loss with elevated temperature due to the thermal degradation of alginate-chitosan and Gn. There are two notable temperature ranges in the degradation of the beads. In the first phase, occurring between 50℃ and 120℃, the Gn-loaded alginate-chitosan beads experienced a weight loss of approximately 9 to 14%, while the alginate-chitosan beads showed a weight loss of around 7.5%, attributable to moisture removal. The second phase was observed between 180℃ and 320℃ with substantial weight loss, representing the fragmentation of polysaccharide and alginate backbone and gentamicin thermal decomposition of the beads (14). A 37.2~41.8% weight loss was observed for the 0.003% Gn-loaded alginate-chitosan beads and 41.5~42.2% (approximately) for the 0.15% Gn-loaded beads until the temperature reached 320℃. Meanwhile, the 0.3% Gn-loaded alginate-chitosan beads experienced a weight decrease of 46.2~55.6%. Notably, increased Gn concentration in the beads was associated with a concurrent increase in weight loss. Once the temperature reached 800℃, the total weight loss observed in the Gn-unloaded and Gn-loaded alginate-chitosan beads was similar, ranging from approximately 67% to 73%, indicating that the residual char content was comparable regardless of gentamicin loading (15, 16).
2. Swelling behavior
Figure 4 illustrates the swelling ratio and PBS uptake of Gn-loaded alginate-chitosan (labeled as xCh-yGn; x = 0.1, 0.5, 1.0; y = 0.003, 0.15, 0.3) beads with different percentages of chitosan (x) and gentamicin (y) as a function of immersion time in PBS solution. The swelling ratio (SR) of the Gn-loaded alginate-chitosan beads was measured for 150 min (Figure 4a). The SR increased steadily, reaching equilibrium at ~90 min, and the weight of the Gn unloaded-alginate-chitosan beads after 150 min ranged from 3228% (0.1Ch) to 3793% (1.0Ch) of the weight of freeze-dried beads. The SR increased with chitosan content ranging from 0.1% to 1.0% in the beads. Comparatively, the SRs of the 0.003% Gn-loaded alginate-chitosan beads were slightly lower than those of unloaded beads. After 150 min, the SR of the Gn-loaded alginate-chitosan beads decreased in the following order: 0.003% Gn (3341% to 3467%), 0.15% Gn (2091% to 2288%), and 0.3% Gn (1751 to 2104%).
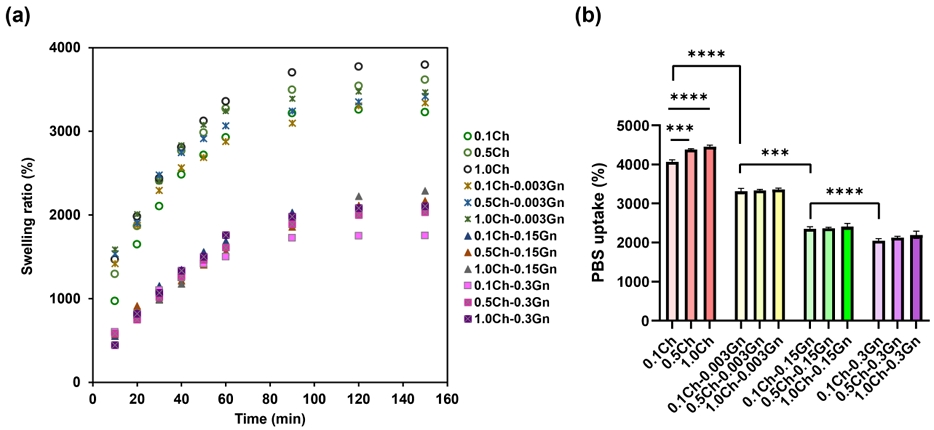
(a) Swelling ratio of gentamicin-loaded alginate-chitosan beads (xCh-yGn) with varying chitosan (Ch; x = 0.1, 0.5, 1.0%) and gentamicin (Gn; y = 0.003, 0.15, 0.3%) concentrations as a function of immersion time in PBS solution. (b) Uptake of PBS to various gentamicin-loaded alginate-chitosan beads after 24 h immersion. Measurements were performed in triplicate. *** p<0.001, **** p<0.0001
Figure 4b shows the PBS uptake of the beads after 24 h. The PBS uptake degree of the 0.3% Gn-loaded alginate-chitosan beads (2142%) was lower than those of 0.003% Gn-loaded (3335%) and 0.15% Gn-loaded (2384%) alginate-chitosan beads.
3. In vitro drug release
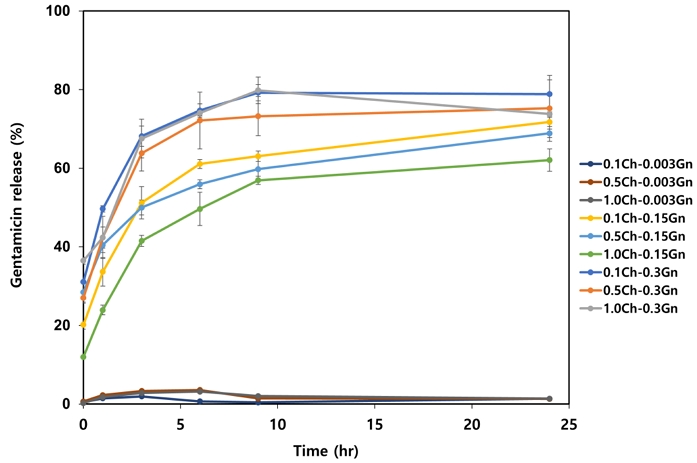
Figure 5 shows the behavior of Gn release from various Gn-loaded alginate-chitosan (xCh-yGn; x = 0.1, 0.5, 1.0 and y = 0.003, 0.15, 0.3) beads with different content of chitosan (x) and gentamicin (y) over a 24 h period. In phosphate-buffered saline solution, drug release behavior was influenced by adjusting the formulation of the alginate-chitosan beads with chitosan and Gn content. The cumulative released amounts gradually increased and reached a plateau after 9 h. For 0.3Gn samples, cumulative release was 73.2% (1.0Ch–0.3Gn) and 79.8% (0.1Ch–0.3Gn), indicating higher release at lower chitosan content. The 0.003Gn alginate-chitosan beads showed the lowest released amount (under 2%) of Gn, whereas 0.15Gn alginate-chitosan beads showed 50~60% release values.
4. Antibacterial activity of extracted gentamicin from beads
Figure 6 illustrates the antibacterial efficacy of fabricated Gn-loaded alginate-chitosan beads against S. aureus (ATCC 29213). Bacterial viability (%) was assessed after 24 h of incubation, with untreated bacterial cultures serving as control (Ctrl, 100% viability). Even Gn-unloaded beads exhibited antibacterial activity, reducing viability by ~50% across all chitosan concentrations. Gn-loaded beads achieved marked inhibition: ~90% (0.003% Gn), ~95% (0.15% Gn), and nearly complete inhibition (0.3% Gn).
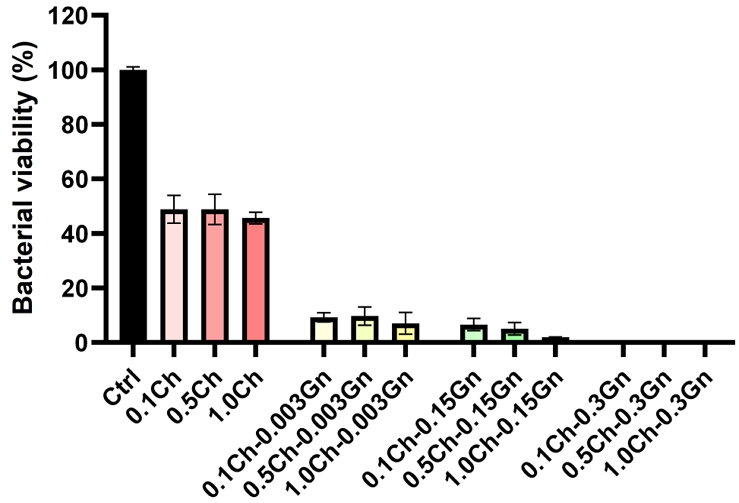
Antibacterial activity of Gn-loaded alginate-chitosan beads (xCh-yGn) with varying chitosan (Ch; x = 0.1, 0.5, 1.0%) and gentamicin (Gn; y = 0.003, 0.15, 0.3%) concentrations against Staphylococcus aureus (ATCC 29213). Bacterial viability (%) was measured after 24 h of incubation. Ctrl: untreated control.
5. Evaluation of cytotoxicity of extracted gentamicin from beads
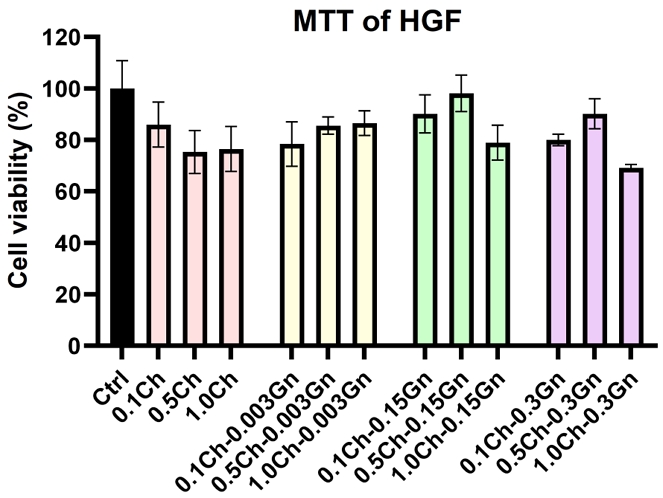
The cytotoxicity of Gn-loaded alginate-chitosan beads on HGF cells after 24 h of the culture period is illustrated in Figure 7. All alginate–chitosan bead groups exhibited >70% cell viability for the HGF cell line. Incorporation of Gn maintained cell viability above 77% in all groups, except 1.0Ch-0.3Gn, demonstrating good biocompatibility. The 1.0Ch-0.3Gn group exhibited mild cytotoxicity, with viability reduced to ~69%.
Discussion
Figure 1 shows SEM images of freeze-dried alginate-chitosan beads fabricated with the compositional variations in alginate, chitosan, and gentamicin concentrations. It illustrates the typical surface morphology of these beads, highlighting that the freeze-drying process resulted in spherical shapes with wrinkled surfaces due to gradual water loss, leading to a collapsed and heterogeneous surface. Consequently, the volume change was not uniform across the system, causing morphological alterations. Understanding how chitosan and Gn content influence bead structure is crucial for optimizing their use in biomedical applications, including drug delivery and tissue engineering. The observed morphologies are also valuable for predicting drug encapsulation efficiency and release performance.
The swelling behavior is crucial in characterizing the beads as it directly affects the release kinetics of the loaded Gn. The swelling behavior and PBS uptake of Gn-loaded alginate-chitosan beads were shown in Figure 4. The SR increased steadily for all formulations, reaching equilibrium at ~90 min.
After 150 min, the SR for Gn-unloaded beads ranged from 3228% (0.1Ch) to 3793% (1.0Ch), indicating that higher chitosan content promoted greater swelling. This increased water uptake likely results from the protonation of primary amino groups (-NH3+) in chitosan, which induces electrostatic repulsion between polymer chains and leads to expansion of the network structure, allowing greater water penetration (17). Conversely, Gn loading reduced SR across all formulations. At 150 min, 0.003% Gn-loaded beads showed SRs of 3341-3467%, slightly lower than the unloaded beads. As the gentamicin concentration increased to 0.15% and 0.3%, swelling decreased more significantly, with SRs ranging from 2091-2288%, and 1751-2104%, respectively. This reduction is likely reflects strong ionic interactions between carboxyl groups in alginates and amino groups in both chitosan and gentamicin, producing denser crosslinking within the bead matrix that restricts water diffusion (18).
Figure 5 demonstrates the effect of chitosan and gentamicin content on drug release kinetics in PBS, showing cumulative release profiles of Gn from alginate-chitosan beads (xCh-yGn; x = 0.1, 0.5, 1.0; y = 0.003, 0.15, 0.3) over a 24 h period. Drug release kinetics were strongly influenced by both chitosan and gentamicin content in the bead formulations. All groups exhibited a rapid initial release, followed by a plateau at ~9 h, indicating a diffusion-controlled release process. The observed release behavior was likely governed by an initial swelling of the hydrogel matrix, followed by partial erosion or disintegration, with the drug release mechanism shifting from diffusion-dominated to polymer relaxation (19). Overall, lower chitosan content was associated with greater Gn release. These results demonstrate that differences in surface morphology and electrostatic interaction among chitosan, alginate, and Gn may contribute to the formulation-dependent drug release efficiency.
This study evaluated the antibacterial efficacy of fabricated Gn-loaded alginate-chitosan beads with varying chitosan and Gn content against S. aureus. As shown in Figure 6, even in the absence of gentamicin, alginate-chitosan beads demonstrated moderate antibacterial activity, reducing bacterial viability by ~50% at all chitosan concentrations (0.1%, 0.5%, and 1.0%), confirming the intrinsic antimicrobial properties of chitosan (7, 12).
Gn loading further enhanced antibacterial activity in a dose-dependent manner. Beads containing 0.003%, 0.15%, and 0.3% Gn inhibited S. aureus growth by ~90%, 95%, and 100%, respectively (p<0.05). This observation highlights synergistic effect between chitosan and Gn. Chitosan exerts anti-bacterial action via membrane disruption and its polycationic charge (13), whereas Gn inhibits bacterial protein synthesis by binding to the 30S ribosomal subunit, resulting in nonfunctional protein production (2, 3). These combined effects compromise metabolic function and structural integrity. Due to the absence of an outer membrane and the presence of a thick peptidoglycan layer (20–40 nm), S. aureus is particularly vulnerable to gentamicin (Gn), which may compromise cell wall integrity, thereby inhibiting bacterial growth and enhancing its overall antibacterial effect (20).
Figure 7 illustrates the cell viability of HGFs after 24 h of incubation with Gn extracts from Gn-loaded alginate-chitosan beads. The groups represented for the beads with various formulations of alginate-chitosan and gentamicin content (xCh-yGn; x = 0.1, 0.5, 1.0% and y = 0.003, 0.15, 0.3%). No cytotoxic effects were observed in the 0.1Ch and 0.5Ch groups, even at the highest Gn concentration (0.3%). However, the combination of high chitosan (1.0%) and high gentamicin (0.3%) content reduced cell viability, suggesting a synergistic effect that may impair cell function. This decrease may be attributed to increased Gn release, which induces cell apoptosis by damaging cell membranes and disrupting metabolic activities (21).
In this study, antibacterial efficacy was assessed against a single species. Future research should expand the spectrum of tested organisms and optimize formulations for enhanced loading and controlled release of multiple therapeutics. Despite these limitations, our findings suggest that Gn-loaded alginate–chitosan beads are promising carriers for localized, sustained release of antimicrobial agents and bioactive factors to support tissue regeneration. Consistent with current findings, natural polymer–based DDS are expected to become more controllable, specific, efficient, and biocompatible. When combined with mineral fillers, these beads could be applied to fill osseous defects after periapical surgery or cyst enucleation/curettage, suppress infection during osteogenesis, and gradually resorb in vivo, supporting defect resolution and new bone formation. Although this study provides a fundamental understanding of composition–property relationships in Gn-loaded alginate–chitosan carriers, the development of more precisely controllable natural polymer-based systems is warranted to maximize their potential as localized therapeutic delivery platforms for oral and maxillofacial applications.
Conclusion
Gn-loaded alginate-chitosan beads were successfully fabricated with varying chitosan and Gn concentrations using ionic gelation method. Increasing the chitosan content from 0.1 to 1.0% in Gn-loaded alginate-chitosan beads induced a transition to smaller, denser, and more wrinkled bead surfaces. Drug release behavior was influenced by both surface morphology and electrostatic interactions among chitosan, alginate, and Gn conjugates. The swelling ratio of the beads increased steadily, reaching equilibrium at ~90 min. PBS uptake rose with increasing chitosan content but decreased with higher Gn loading. Adjusting chitosan and Gn concentrations allowed precise modulation of drug release kinetics. The release Gn showed >90% antibacterial efficacy against S. aureus, and all bead formulations showed were cytocompatible (>77% viability in HGF cells) except for the 1.0Ch-0.3Gn group, which showed mild cytotoxicity. Overall, these results highlight the potential of compositionally tunable alginate-chitosan beads as biocompatible platforms for localized antimicrobial drug delivery in biomedical applications.
Acknowledgments
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (NRF-2019R1A5A2027521) and Ministry of Education (No. NRF-2021R1I1A1A01055994).
References
-
Alamoush RA, Kushnerev E, Yates JM, Satterthwaite JD, Silikas N. Response of two gingival cell lines to CAD/CAM composite blocks. Dent Mater. 2020;36(9):1214–25.
[https://doi.org/10.1016/j.dental.2020.05.014]

-
Nayak AK, Hasnain MS, Aminabhavi TM. Drug delivery using interpenetrating polymeric networks of natural polymers: A recent update. J Drug Deliv Sci Technol. 2021;66:102915.
[https://doi.org/10.1016/j.jddst.2021.102915]

-
Xu Y, Zhan C, Fan L, Wang L, Zheng H. Preparation of dual crosslinked alginate-chitosan blend gel beads and in vitro controlled release in oral site-specific drug delivery system. Int J Pharm. 2007;336:329–37.
[https://doi.org/10.1016/j.ijpharm.2006.12.019]

-
Cheng CH, Chen YS, Chang HT, Chang KC, Huang SM, Liu SM, Chen WC. In vitro evaluation of antibacterial activity and biocompatibility of synergistically cross-linked gelatin-alginate hydrogel beads as gentamicin carriers. J Drug Deliv Sci Technol. 2023;79:104078.
[https://doi.org/10.1016/j.jddst.2022.104078]

-
Wang Y, Wang W, Shi X, Wang A. Enhanced swelling and responsive properties of an alginate-based superabsorbent hydrogel by sodium p-styrenesulfonate and attapulgite nanorods. Polym Bull. 2013;70:1181–93.
[https://doi.org/10.1007/s00289-012-0901-0]

-
Nagarwal RC, Kumar R, Pandit J. Chitosan coated sodium alginate-chitosan nanoparticles loaded with 5-FU for ocular delivery: In vitro characterization and in vivo study in rabbit eye. Eur J Pharm Sci. 2012;47:678–85.
[https://doi.org/10.1016/j.ejps.2012.08.008]

-
Bakhsheshi-Rad HR, Hadisi Z, Ismail AF, Aziz M, Akbari M, Berto F, Chen X. In vitro and in vivo evaluation of chitosan-alginate/gentamicin wound dressing nanofibrous with high antibacterial performance. Polym Test. 2020;82:106298.
[https://doi.org/10.1016/j.polymertesting.2019.106298]

-
Grant GT, Morris ER, Rees DA, Smith PJC, Thom D. Biological interactions between polysaccharides and divalent cations: the egg-box model. FEBS Lett. 1973;32(1):195–8.
[https://doi.org/10.1016/0014-5793(73)80770-7]

-
Tong X, Pan W, Su T, Zhang M, Dong W, Qi X. Recent advances in natural polymer-based drug delivery systems. React Funct Polym. 2020;148:104501.
[https://doi.org/10.1016/j.reactfunctpolym.2020.104501]

-
Popov S, Paderin N, Khramova D, Kvashninova E, Patova O, Vityazev F. Swelling, protein adsorption, and biocompatibility in vitro of gel beads prepared from pectin of hogweed Heracleum sosnowskyi Manden in comparison with gel beads from apple pectin. Int J Mol Sci. 2022;23:3388.
[https://doi.org/10.3390/ijms23063388]

-
Bonafé ACF, Oliveira DFLM, Fernandes EE, Garcia MT, Dias IPSS, Bressane A, de Oliveira W, de Mello Rode S. Microbiological evaluation in invisible aligner chemical cleaning methods against Candida albicans and Streptococcus mutans. Am J Orthod Dentofacial Orthop. 2023;164:e43–50.
[https://doi.org/10.1016/j.ajodo.2023.05.014]

-
Lawrie G, Keen I, Drew B, Chandler-Temple A, Rintoul L, Fredericks P, Grøndahl L. Interactions between alginate and chitosan biopolymers characterized using FTIR and XPS. Biomacromolecules. 2007;8:2533–41.
[https://doi.org/10.1021/bm070014y]

-
Li W, Li W, Wan Y, Wang L, Zhou T. Preparation, characterization and releasing property of antibacterial nano-capsules composed of ε-PL-EGCG and sodium alginate-chitosan. Int J Biol Macromol. 2022;204:652–60.
[https://doi.org/10.1016/j.ijbiomac.2022.01.123]

-
Yasir N, Khan AS, Hassan MF, Ibrahim TH, Khamis MI, Nancarrow P. Ionic liquid agar-alginate beads as a sustainable phenol adsorbent. Polymers. 2022;14(5):984.
[https://doi.org/10.3390/polym14050984]

-
Park KH, Choi YW, Ryu HJ, Lee HJ, Moon JH, Song HJ, Park YJ. Controlled drug release using chitosan-alginate-gentamicin multi-component beads. Materials. 2022;15(21):7682.
[https://doi.org/10.3390/ma15217682]

-
Sadeghianmaryan A, Naghieh S, Yazdanpanah Z, Sardroud HA, Sharma N, Wilson LD, Chen X. Fabrication of chitosan/alginate/hydroxyapatite hybrid scaffolds using 3D printing and impregnating techniques for potential cartilage regeneration. Int J Biol Macromol. 2022;204:62–75.
[https://doi.org/10.1016/j.ijbiomac.2022.01.201]

- Dolatabadi-Farahani T, Vasheghani-Farahani E, Mirzadeh H. Swelling behaviour of alginate–N,O-carboxymethyl chitosan gel beads coated by chitosan. Iran Polym J. 2006;15(5):405–15.
- Gierszewska M, Ostrowska-Czubenko J. Effect of ionic crosslinking agents on swelling behaviour of chitosan hydrogel membranes. Prog Chem Appl Chitin Its Deriv. 2016;21:25–34.
-
Varma MV, Kaushal AM, Garg A, Garg S. Factors affecting mechanism and kinetics of drug release from matrix-based oral controlled drug delivery systems. Am J Drug Deliv. 2004;2:43–57.
[https://doi.org/10.2165/00137696-200402010-00003]

-
Barzic AI, Ioan S. Antibacterial drugs-From basic concepts to complex therapeutic mechanisms of polymer systems. In: Bobbarala V, editor. Conceps, compounds and the alternatives of antibacterials. London: IntechOpen; 2015.
[https://doi.org/10.5772/60755]

-
He G, Chen X, Yin Y, Cai W, Ke W, Kong Y, Zheng H. Preparation and antibacterial properties of O-carboxymethyl chitosan/lincomycin hydrogels. J Biomater Sci Polym Ed. 2016;27:370–84.
[https://doi.org/10.1080/09205063.2015.1132616]