Effect of polymer-coupled Cu-TiO2 nanoparticles on the bleaching of stained teeth under hydrogen peroxide and blue light irradiation
Abstract
Everybody wishes to have clean and white teeth. The purpose of the present study was to investigate the effect of polymer-coupled-copper-doped titanium dioxide (Cu-TiO2) nanoparticles (NPs) on the bleaching of stained teeth under treatment of hydrogen peroxide (H2O2; HP) and blue light irradiation together. For the study, Cu and TiO2 were mixed and coupled using polymer to form polymer-coupled Cu-TiO2 (PCT). The light absorbance of PCT was measured. To evaluate the possibility of radical production, methylene blue (MB) degradation and terephthalic acid (TA) assays were performed under various conditions. Color changes in the stained teeth including the absence or presence of 3% HP and light irradiation were monitored. The cell toxicity of PCT on normal cells was evaluated, and the cell morphology of the treated was observed after cell viability test. Consequently, PCT exhibited a different absorbance in the 600-800 nm range, compared to the pure TiO2, depending on the content of incorporated Cu. The greatest MB degradation and increase of fluorescence peak near 425 nm were spectroscopically observed for the combined treatment of PCT NPS, HP, and blue light, which indicates the production of radicals with time. Bleaching of the stained teeth by the PCT NPs + HP + laser condition was significantly different compared to the rest conditions, including 15% HP alone, during the same treatment time.
초록
깨끗하고 흰 치아를 가지는 것은 많은 사람들의 오랜 바람이다. 본 연구는 폴리머-구리-이산화티타늄 나노입자에 과산화수소와 파란 빛을 함께 적용했을 때 착색된 치아에 나타나는 미백효과를 평가한 것이다. 이를 위하여 Cu가 도핑된 Cu-TiO2를 만들고 다시 폴리머가 결합된 폴리머-Cu-TiO2 (PCT) 나노입자를 합성하였다. 이후에 이들의 흡광도를 측정하고, 라디칼 생성 여부를 평가하기 위해서 methylene blue (MB) degradation과 terephthalic acid (TA) assay를 하였다. 착색된 치아의 색 변화를 3% hydrogen peroxide (HP)와 광조사 유무 조건하에서 실시하였다. 그리고 PCT 나노입자의 독성 여부를 cell viability test로 평가하고 이후에 cell의 상태를 현미경으로 관찰하였다. 그 결과 PCT 나노입자의 흡광도는 600-800 nm에서 순수한 TiO2와 달랐고 이는 투입된 Cu의 양과 상관이 있었다. PCT 나노입자 + HP + 광조사 조건에서 MB degradation과 425 nm근처에서의 형광강도가 가장 강했는데 이는 라디칼이 시간에 따라 생성되는 것과 상관이 있었다. 착색된 치아에 대해서 이 조건을 적용했을 때 가장 크고 의미 있는 색변화가 있었는데 동일한 시간동안 반응했을 때 15% HP 단독에 의한 색 변화보다도 더 큰 변화가 있었다. 이로써 PCT 나노입자는 HP와 광조사를 함께 적용했을 때 착색된 치아에 효과적인 미백을 기대할 수 있다.
Keywords:
Stained teeth, Bleaching, Polymer-Cu-TiO2키워드:
착색된 치아, 미백, 폴리머-Cu-TiO2Introduction
The enduring public desire for clean and white teeth has established tooth bleaching (whitening) as a significant concern within contemporary dentistry (1-3). Although primarily an aesthetic procedure rather than a medical necessity, the demand for brighter smiles remains consistently high. However, achieving and maintaining optimal tooth color is challenging due to numerous intrinsic and extrinsic risk factors. Common culprits include aging, smoking, and the regular consumption of chromogenic foods and beverages. When stains from these sources are adsorbed onto the tooth surface and subsurface through prolonged and repeated exposure, they can lead to undesirable discoloration if not adequately removed. While daily tooth brushing offers a simple primary measure for stain removal, it is often insufficient for eliminating heavy, persistent stains, making some degree of color change inevitable over time.
Hydrogen peroxide (HP; H2O2) and carbamide peroxide (CP; CH6N2O3) are the most widely employed bleaching agents in both home-use and clinical settings. HP is readily available in low concentrations (typically up to 3%) as an over-the-counter product, while higher concentrations (e.g., <16%) are professionally administered in clinics. The efficacy of HP primarily stems from its ability to produce reactive oxygen species, particularly hydroxyl radicals (OH•), which are potent active components for breaking down stains. OH• can be generated by various methods, including heat, ultraviolet (UV) light, or catalysts such as transition metals (e.g., iron, copper, or manganese) (4-7). These strong oxidizing agents degrade the long, complex molecules of stains into smaller, colorless molecules that no longer absorb and reflect light in the visible spectrum. Despite the proven efficacy and convenience of HP and CP, the bleaching process can unfortunately lead to side effects such as temporary tooth sensitivity, mineral loss, and dehydration of dental tissues (8, 9). Therefore, developing methods to achieve satisfactory bleaching outcomes while minimizing adverse effects through lower concentrations and shorter treatment times is a critical objective. To address these limitations, various non-peroxide alternatives have also been explored, including enzymes that break down protein-based stains and abrasive agents like baking soda for surface stain removal (10-12). While some of these alternatives show promise, their overall efficacy and safety profiles can vary significantly between products.
Titanium dioxide (TiO2) presents itself as a valuable material with potential for enhancing tooth bleaching due to its photocatalytic properties. Photocatalysis is a light-induced process where light energy activates a catalyst (commonly TiO2) to accelerate chemical reactions. Upon irradiation with light possessing photon energy greater than its bandgap, TiO2 generates electron-hole pairs. The holes in the valence band can then produce OH• by reacting with environmental water or by oxidizing adsorbed HP (13-15). Furthermore, modifying the surface of TiO2 can improve light absorption beyond 400 nm. Specific doped metals like copper (Cu) or magnesium (Mg) can induce a Fenton-like reaction with HP, further enhancing OH• generation. The Fenton-like reaction is an oxidation process using metal catalysts other than, or in addition to, ferrous iron (Fe2+) which catalyzes the decomposition of HP in acidic conditions to generate highly reactive OH•. Consequently, integrating TiO2, or its modified forms, with light irradiation holds the potential to augment the efficacy of HP alone, thereby allowing for reduced HP concentrations and shorter overall treatment durations. This approach could mitigate the risks associated with conventional high-concentration peroxide treatments. The present study aimed to evaluate the enhanced color change of stained teeth using PCT nanoparticles (NPs) in conjunction with blue light irradiation to actively boost the bleaching reaction. metal catalysts other than, or in addition to, ferrous iron.
Materials and Methods
1. Synthesis of polymer-coupled Cu-TiO2 NPs
Commercial pristine TiO2 was used to synthesize polymer-coupled Cu-TiO2 NPs. First, 100 mg of polymer, polyvinylpyrrolidone (pvp), was added to 10 mL of distilled water (dw), then 100 mg of TiO2 was added and sonicated for 30 minutes. After that, different amount (25, 50, 100, 150 mg) of CuCl2∙2H2O was added and stirred for 2 hours. Obtained products were fully dried and ground finely using a mortar for next use.
2. Absorbance of pvp-Cu-TiO2 (PCT) NPs
Absorbance of the used NPs was measured using a spectrophotometer (SpectraMax 190, Molecular Devices, San Jose, CA, USA).
3. Methylene blue (MB) degradation test
PCT NPs were used to test MB degradation with different conditions if it can perform photocatalytic reaction under light irradiation. To perform the test, 4 mL MB solution (10 ppm) was mixed with PCT NPs (1 wt%) in a 5 mL vial, and stirred using a magnetic bar during light irradiation. For light irradiation, 405 nm laser was used at 50 mW/cm2 intensity for 10 minutes. After that, the color of MB solution was measured using the same spectrophotometer. HP of 3% and 15% was applied to compare the outcomes with those of PCT NPs. All the reaction in MB solution was continued for 30 minutes including light irradiation.
4. Terephthalic acid (TA) assay
The Fenton-like activity of PCT NPs was evaluated using TA fluorescence assay. Test solutions were prepared as follows: At first, 2 mM TA and 5 mM NaOH were mixed, then the mixed solution was combined with dw, 0.3 mL TA-NaOH, 200 ppm of PCT NPs, and 0.15% H2O2. The fluorescence of the test solution was measured at different time points of laser irradiation using a spectrofluorometer (Cary Eclipse, Agilent, Santa Clara, CA, USA).
5. Color difference measurement using stained teeth
To evaluate the teeth bleaching, color change of the stained teeth was measured. For the study, caries-free sound teeth (5 teeth for each condition) were stained using coffee solution to achieve uniform stain on the tooth surface after immersing for 1 month. To make the coffee solution, 0.9 g of black coffee powder was mixed with 100 mL hot water. The coffee solution was replaced every two days. To determine the initial color values before bleaching, the first color measurements were performed using a digital spectrophotometer (VITA Easyshade V, VITA Zahnfabrik, Bad Säckingen, Germany) following the instruction. After the initial color measurement, all teeth surfaces were treated with a test agent, followed by laser irradiation for 3 hours with 50 mW/cm2 intensity. After that, the treated surface was cleaned using tap water, cleared remaining water using soft tissue, and the second color measurement was performed as before at the first measured position. After that, using the obtained color values (L*, a*, b*), the color difference was calculated using the formula:
- ΔE* = [ (ΔL*)2 + (Δa*)2 + (Δb*)2 ]1/2
where ΔL*, Δa*, and Δb* are the changes in L*, a*, and b*, respectively.
6. Cell viability test
To assess cell viability, cells (HEK293; Human embryonic kidney cells) were seeded in 96-well plates (1×104 cells/well) and incubated for 12 hours before treatment. The cells were then treated with PCT NPs at the desired concentrations for 24 hours. The culture medium was replaced with fresh medium containing 10% CCK-8 solution and incubated for 1 hour. The absorbance of the cells in each well was measured at 450 nm using a microplate reader (BioTek Synergy HTX, Agilent, Santa Clara, CA, USA). After the cell viability test, the cells were observed using an optical microscope (Nikon; Eclipse Ts2, Tokyo, Japan).
7. Statistical analysis
The result of color differences was analyzed by one-way ANOVA (SPSS) followed by a Tukey’s post-hoc test for multiple comparisons; p values <0.05 were considered significant.
Results
1. UV-Vis absorbance spectra
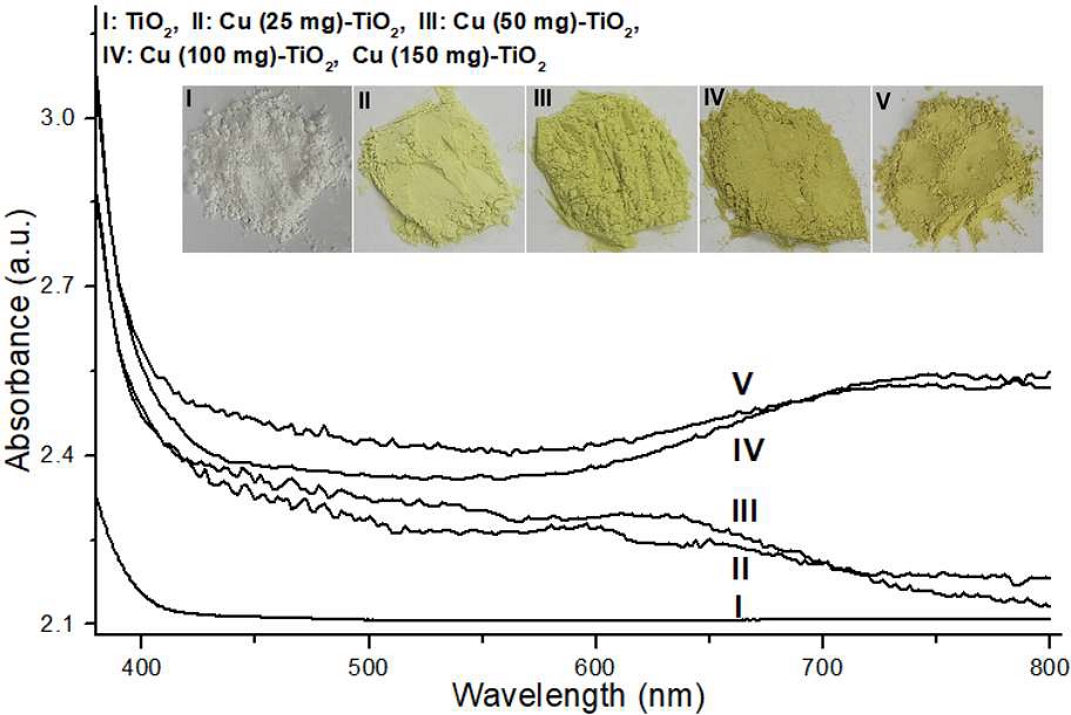
Figure 1 illustrates the UV-Vis absorbance spectra of PCT NPs with varying Cu content, alongside their macroscopic appearance (inset). In contrast to pure TiO2, which exhibited negligible light absorption beyond 400 nm, PCT NPs consistently demonstrated a gradual increase in absorbance intensity after 400 nm. Furthermore, the absorbance profile in the 600-800 nm range showed either an ascending or descending trend, which was directly influenced by the specific Cu content. Consistent with these spectroscopic observations, the visual appearance of the PCT NPs, as depicted in the inset, progressively shifted from pale yellow to dark yellow with increasing Cu incorporation.
2. MB degradation
The degradation efficiency of methylene blue (MB) solution under various treatment conditions is presented in Figure 2. Treatment of the 10 ppm MB solution with HP alone resulted in a minor degradation of up to 15%. In distinct contrast, the incorporation of PCT NPs with HP significantly enhanced MB degradation, leading to the complete transparency of the solution following light irradiation.
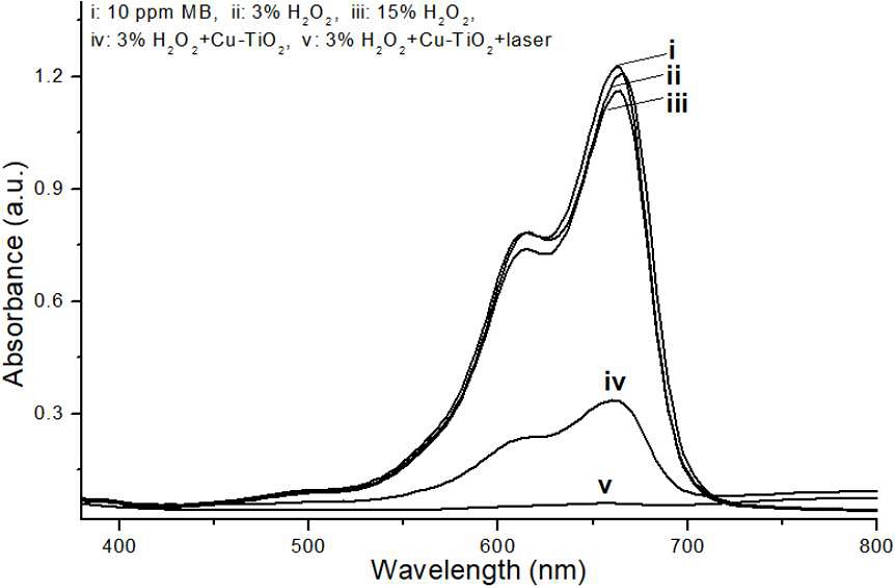
MB degradation under various treatment conditions. The graph illustrates the extent of MB degradation for a 10 ppm MB solution under different experimental conditions. Significant degradation of the initial MB solution was observed when treated with a combination of PCT NPs and HP (condition iv). This degradation was further enhanced, leading to complete transparency of the solution, upon additional blue light irradiation (condition v). In contrast, weak MB degradation was observed with HP (3% or 15%) alone.
3. TA assay
The outcomes of the TA assay are presented in Figure 3. Assay solutions treated with only dw or solely with PCT NPs exhibited either no detectable or only barely discernible fluorescence. In apparent contrast, the condition combining PCT NPs with HP under light irradiation demonstrated the highest fluorescence intensity, specifically observed near 425 nm. This fluorescence signal is consistent with a significant production of OH• under this specific treatment regimen.
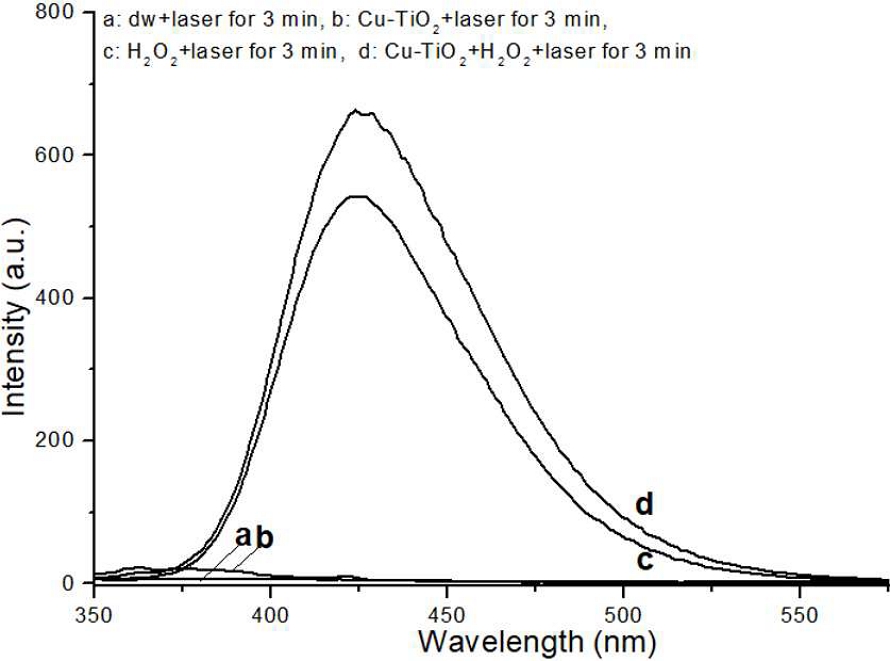
TA assay results for OH• detection. The graph displays the fluorescence intensity measured from TA assay solutions under light irradiation. A significant increase in fluorescence intensity near 425 nm was observed in solutions containing either HP alone or a combination of PCT NPs and HP. This notable fluorescence indicates significant production of OH• under light irradiation.
4. Color difference
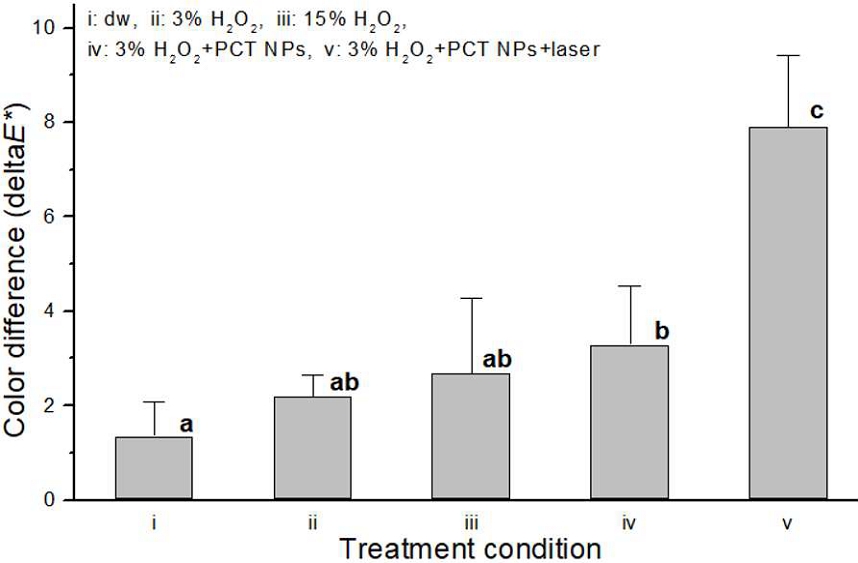
The color difference (ΔE*) of stained teeth under various treatment conditions is presented in Figure 4. Among all tested conditions, condition v, involving PCT NPs combined with 3% HP and 405 nm laser irradiation, yielded the greatest color difference (ΔE* = 7.9 ± 1.5). This observed color difference was statistically significant (p<0.05) when compared to all other tested conditions.
5. Cell viability
Figure 5 presents the findings from the cell viability these PCT NPs much enhanced the efficacy assay and observations of cellular morphology following treatment. Specifically, normal HEK293 cells treated with PCT NPs exhibited no adverse effects on viability at concentrations up to 500 ppm (Figure 5A). Furthermore, the morphology of these treated cells remained consistent with that of untreated control cells (Figure 5B and 5C). These morphological findings are in strong agreement with the quantitative cell viability outcomes.
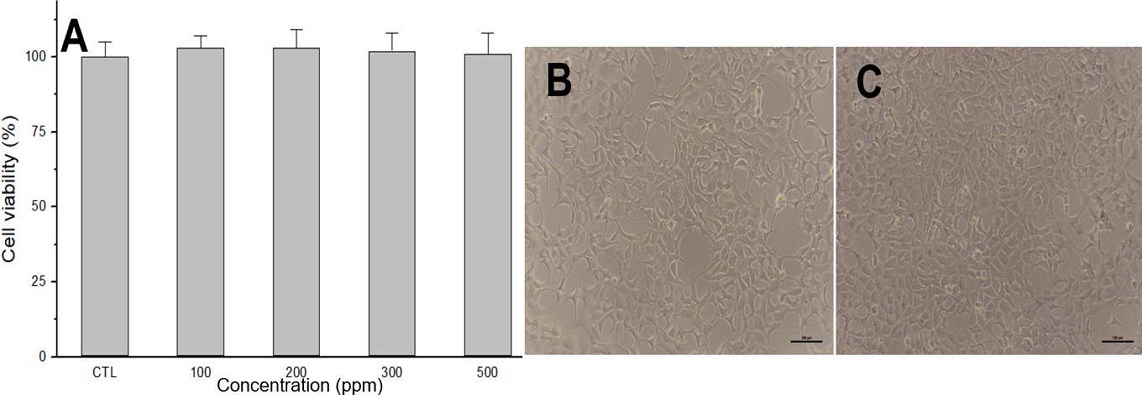
Cell viability and morphological assessment of HEK293 cells after PCT NPs treatment. (A) Results from the cell viability test demonstrate that normal HEK293 cells remained unaffected by PCT NPs at concentrations up to 500 ppm. (B) Representative image of untreated HEK293 cells. (C) HEK293 cells treated with PCT NPs show no visible changes in morphology compared to the untreated control (B).
Discussion
Tooth discoloration is a common aesthetic concern driven by daily exposure to various extrinsic factors like foods, beverages, and smoking, leading to the accumulation of chromophores on and within the tooth structure. While regular brushing helps to mitigate this, residual and gradually accumulating stains often necessitate further intervention. Peroxide-based teeth bleaching remains a widely adopted and accessible method for addressing such discoloration.
Conventional peroxide-based bleaching primarily utilizes HP or CP at varying concentrations, tailored for either at-home or in-office applications. At-home bleaching typically involves low concentration 3% HP, readily available over-the-counter, requiring prolonged treatment durations of 6-8 hours daily over several weeks. In contrast, dental clinics employ higher concentrations of HP (<15%) under professional supervision, significantly reducing treatment time to less than 0.5 hours per session over 1-2 weeks. While higher HP concentrations offer faster and more noticeable whitening, they are frequently associated with undesirable side effects, including acute tooth sensitivity or deep pain, mineral loss, and dehydration. These concerns underscore the need for safer yet equally effective bleaching alternatives, particularly those that can achieve comparable or superior results with lower peroxide concentrations, thereby reducing potential adverse effects and overall treatment time (16, 17).
The present study introduces an effective approach to peroxide-based teeth bleaching by combining HP with photocatalytic titanium dioxide nanoparticles (PCT NPs) under blue light irradiation. The enhanced efficacy of this system is attributed to the synergistic production of reactive oxygen species (ROS), primarily OH•, through Fenton-like and photocatalytic reactions involving HP, PCT NPs, and blue light. The increased radical generation was quantitatively confirmed by the enhanced degradation of MB solution when HP + PCT NPs + laser was compared to HP+PCT NPs alone, highlighting the significant contribution of light activation. Furthermore, the 2-hydroxyterephthalic acid (HTA) fluorescence assay, which detects OH•, unequivocally demonstrated a substantial increase in OH• production in the presence of HP + PCT NPs + laser compared to HP+laser, firmly establishing OH• as the primary drivers of the observed bleaching effect (18, 19).
The practical implications of the present study demonstrate that utilizing PCT NPs, HP, and blue light irradiation achieves superior bleaching efficacy compared to treatment with 15% HP alone, even within the same treatment period. This advancement suggests a favorable pathway for achieving substantial whitening with much lower HP concentrations (e.g., 3% HP), potentially outperforming traditional high-concentration HP treatments. Such a system, particularly when employing Cu-combined-TiO2-based NPs under blue light, could significantly reduce the total treatment duration. Crucially, by enabling effective bleaching with lower HP concentrations and shorter treatment times, the tested method in the present is expected to minimize common drawbacks associated with conventional high-concentration HP treatments, such as tooth pain, mineral loss, and dehydration. Moreover, the safety profile of the tested PCT NPs is highly favorable, showing negligible toxicity to normal HEK293 cells up to 500 ppm concentrations and maintaining normal cellular morphology after the bleaching tests, thus addressing potential safety concerns regarding nanoparticle usage. To achieve the current result, laser treatment was applied for 3 hours, which is too long for a patient sitting in a chair. Therefore, future studies should consider significantly reducing both the irradiation time and light intensity to have clinical implication.
Conclusion
Within the limit of the study, the tested PCT NPs exhibited excellent biocompatibility, showing safety for normal cells at concentrations up to 500 ppm. When integrated with 3% HP and subjected to blue light irradiation, PCT NPs significantly enhanced the efficacy of tooth bleaching in stained teeth, achieving superior results compared to treatment with 15% H2O2 alone within the same duration. The underlying mechanism for this augmented bleaching effect is primarily attributed to the production of OH•, a process further boosted by the combined application of HP and blue light irradiation. These findings collectively establish the integrated application of PCT NPs with HP and blue light irradiation as a highly effective and promising strategy for advanced tooth bleaching. Further investigations are warranted to explore its full clinical potential and applications.
References
-
Hattab FN. Tooth Discoloration: Causes and Clinical Presentation-Part I. J Oral Health Comm Dent. 2024;18: 63-74.
[https://doi.org/10.5005/jp-journals-10062-0187]

-
Joiner A, Luo W. Tooth colour and whiteness: A review. J Dent. 2017;67S:S3-S10.
[https://doi.org/10.1016/j.jdent.2017.09.006]

-
Kwon SR, Wertz PW. Review of the Mechanism of Tooth Whitening. J Esthet Restor Dent. 2015;27:240-57.
[https://doi.org/10.1111/jerd.12152]

-
Buchalla W, Attin T. External bleaching therapy with activation by heat, light or laser-a systematic review. Dent Mater. 2007;23:586-96.
[https://doi.org/10.1016/j.dental.2006.03.018]

- Maryline M, René S. Vital tooth bleaching: Biologic adverse effects-A review. Quintessence Int. 2008;39: 645-59.
-
Matthias Epple M, Frederic Meyer F, Enax J. A Critical Review of Modern Concepts for Teeth Whitening. Dent J. 2019;7:79.
[https://doi.org/10.3390/dj7030079]

-
Guo B, Tao Y, Yang T, Su X, Tan X, Tian W, Xie L. Biomaterials based on advanced oxidation processes in tooth whitening: fundamentals, progress, and models. J Mater Chem B. 2024;12:9459-77.
[https://doi.org/10.1039/D4TB01311E]

-
Ruiz-López J, Pulgar R, Lucena C, Pelaez-Cruz P, Cardona JC, Perez MM, Ghinea R. Impact of short-term dental dehydration on in-vivo dental color and whiteness. J Dent. 2021;105:103560.
[https://doi.org/10.1016/j.jdent.2020.103560]

-
Lee KH, Kim HI, Kim KH, Kwon YH. Mineral loss from bovine enamel by a 30% hydrogen peroxide solution. J Oral Rehabil. 2006;33:229-33.
[https://doi.org/10.1111/j.1365-2842.2004.01360.x]

- Efficacy of natural teeth whitening remedies: Pineapple and banana. Lee S, Mercado D, Feceu C, Davalos C, Rahgozar N, Oyoyo U, Kwon SR. Am J Dent. 2025;38:16A-19A.
-
Bizhang M, Domini J, Danesh G, Zimmer S. Effectiveness of a new non-hydrogen peroxide bleaching agent after single use - a double-blind placebo controlled short-term study. J Appl Oral Sci. 2017;25:575-84.
[https://doi.org/10.1590/1678-7757-2016-0463]

- Chan DCN, Cao D. Updates on non-peroxide dental bleaching agents. Am J Dent. 2025;38:32A-37A.
-
Abdullah AM, Gracia-Pinilla MA, Pillai SC, O’Shea K. UV and Visible Light-Driven Production of Hydroxyl Radicals by Reduced Forms of N, F, and P Codoped Titanium Dioxide. Molecules. 2019;24:2147.
[https://doi.org/10.3390/molecules24112147]

-
Tang Z, Zhao P, Wang H, Liu Y, Bu W. Biomedicine Meets Fenton Chemistry. Chem Rev. 2021;121:1981-2019.
[https://doi.org/10.1021/acs.chemrev.0c00977]

-
Guo Q, Zhou C, Ma Z, Yang X. Fundamentals of TiO2 Photocatalysis: Concepts, Mechanisms, and Challenges. Adv Mater. 2019;31:1901997.
[https://doi.org/10.1002/adma.201901997]

-
Zach L, Cohen G. Pulp response to externally applied heat. Oral Surg Oral Med Oral Pathol. 1965;19:515-30.
[https://doi.org/10.1016/0030-4220(65)90015-0]

-
Pontes M, Gomes J, Lemos C, Leão RS, Moraes S, Vasconcelos B, Pellizzer EP. Effect of Bleaching Gel Concentration on Tooth Color and Sensitivity:Systematic Review and Meta-analysis. Oper Dent. 2020;45:265-75.
[https://doi.org/10.2341/17-376-L]

-
Gonzalez DH, Kuang XM, Scott JA, Rocha GO, Paulson SE. Terephthalate Probe for Hydroxyl Radicals: Yield of 2-Hydroxyterephthalic Acid and Transition Metal Interference. Anal Lett. 2018;51:2488-97.
[https://doi.org/10.1080/00032719.2018.1431246]

-
Xia S, Zhang L, Pan G, Qian P, Ni Z. Photocatalytic degradation of methylene blue with a nanocomposite system: synthesis, photocatalysis and degradation pathways. Phys Chem Chem Phys. 2015;17:5345-51.
[https://doi.org/10.1039/C4CP03877K]