
The Color Stability of Resin Composite After Exposure to pH Solutions and Immerse Into Staining Beverages
초록
본 연구는 3가지 다른 pH 용액에 노출된 후 착색 음료에 침전된 복합 레진의 색 변화를 평가하였다. 실험을 위해 A3색을 가진 복합레진 Z350 XT(3M ESPE, St. Paul, MN, USA)으로 직경 8 mm, 두께 2 mm의 시편을 제작하였다. 각 10개의 시편으로 서로 다른 3가지pH용액(pH 3, pH 6, pH 9)에 14일 동안 노출 후 시편들을 다시 3그룹의 착색음료(증류수, 커피, 포도주스)에 3일 동안 침전시켰다. 착색 음료 침전 전과 후의 L*, a*, b*값을 spectrophotometer로 측정하고 측정값으로 색 변화와 투명도의 변화를 계산하였다. 계산된 결과값을 ANOVA와 Tukey’s test로 분석하여 다음과 같은 결과를 얻었다. 실험 결과, 색 변화(ΔE) 값은 착색음료 종류에 따른 통계학적 유의한 차이는 있었으나(p < 0.05), 이전의 노출된 pH 용액에 따른 유의한 차이는 없었다(p > 0.05). 착색 음료 침전 후 반투명도 지수(TP) 값은증류수를 제외하고 감소되는 경향이었고, 반투명도 지수 변화(ΔTP)는 포도주스에서 가장 높았다. 결론적으로 복합 레진의 색 안정성은기존의 다양한 pH 용액에 노출된 상태가 아니라 착색 음료에 의존하는 것으로 사료된다.
Keywords:
Color stability, pH Solution, Staining beverage, Composite resinⅠ. INTRODUCTION
Composite resins are used most frequently in dentistry for aesthetic restorations. They have many advantages in dental practices, such as their ability to bond to the enamel and dentine, resemblance in color and mechanical properties to the tooth structures, ease of chair-side applications and relatively low cost. A major disadvantage of resin composites, however, is their tendency to discolor, which is a major problem in long-term clinical studies (Millar et al., 1997; van Dijken, 2001).
Color stability is considered as one of the most important factors when selecting composite resin materials for aesthetic restorations. Composite resins change their optical properties with time and that internal discolorations often necessitate restoration replacement. Therefore, restorative materials should match the initial tooth shade and preserve the aesthetic semblance over a period of years in the restored tooth.
The color changes in resin composites are caused by intrinsic and extrinsic factors. External discoloration can be caused by colorants contained in beverages, foods through adsorption and absorption as well as by oral habits, such as tobacco use and certain dietary patterns (Asmussen and Hansen, 1986; Bagheri et al., 2005; Lee et al., 2014). In addition, the resin material can be discolored by intrinsic factors, such as alterations of the resin matrix, filler, loading and particle size distributions, and type of photoinitiator discoloring the resin material (Sarafianou et al., 2007; Barutcigil and Yildiz, 2012).
The consumption of certain beverages can affect the esthetic and physical properties of resin composites. Many studies have demonstrated in vitro that common drinks and food ingredients, such as coffee, tea or red wine, fruit juices, and cola drinks, can cause significant changes in the surface color of composite resin materials (Guler et al., 2005; Fujita et al., 2006; Mohan et al., 2008; Ardu et al., 2010). Certain food colorants (e.g. coffee) can cause more severe staining than others (e.g. cola), despite having similar color parameters (Mundim et al., 2010; Barutcigil and Yildiz, 2012). Stober et al.(2001) reported that red wine caused the most severe discoloration compared to tea, coffee, mouth rinse, turmeric solutions and UV irradiation staining agents. The staining of resins by beverages is caused by the adsorption or absorption of colorants by the resins (Ruyter, 1995) and the resin's affinity for extrinsic stains is modulated by its water sorption rate (Ortengren et al., 2001b).
An extensive literature review (Patel et al., 2004; Bagheri et al., 2005; Villalta et al., 2006; Cilli et al., 2012) provided little evidence for the effect of acidic solutions on restorative composite resin materials. A low pH was reported to adversely affect the surface integrity by softening the matrix, causing a loss of structural ions and affecting the wear resistance and discoloration of dental materials (Patel et al., 2004; Bagheri et al., 2005; Villalta et al., 2006). On the other hand, highly alkaline solutions have also been shown to accelerate hydrolysis and produce quick surface micro-structural damage (Cilli et al., 2012).
Discoloration can be assessed visually and using instrumental techniques. Instrumental techniques eliminate the subjective interpretation inherent in a visual color comparison. Spectrophotometers and colorimeters are widely used tools for detecting the color changes in dental restorative materials (Asmussen and Hansen, 1986). Studies have shown that, when observing the color differences in aesthetic restorations, although a value of ΔE > 1 is visually perceptible, ΔE > 3.3 was found to be a critical value for the visual perception of a restoration(Schulze et al., 2003).
Although numerous experimental models have been used to examine the color stability and stain resistance of composite resin materials, few studies have considered the pH environment of the oral cavity during the consumption of colorant-containing beverages and foods. pH variations can affect the surface and structural integrity of composite resin materials, rendering the restorations more susceptible to staining and discoloration (Minami et al., 2007; Valinoti et al., 2008).
Therefore, this study examined the color stability of nanofilled resin composites after exposure to solutions at three different pHs followed by immersion in staining beverages using a reflection spectrophotometer based on the CIE (Commission Internationale de l’Eclairage) L*a*b* color system. The null hypotheses tested were (1) the acidity of pH solution that the resin is exposed to do not influence the color stability of the material, and (2) the type of immersion beverage has no effect on the discoloration susceptibility of the composite resin.
Ⅱ. MATERIALS AND METHODS
1. Specimen preparation
For the study, composite resin (Filtek Z350, 3M ESPE, St.Paul, MN, USA) with the shade, A3, were used. A LED light-curing unit (LCU) (L.E.Demetron, Kerr, Danbury, CT, USA) was used for light curing. To prepare the specimens, a metal ring mold (inner diameter: 8 mm; thickness: 2 mm; n=10 for each condition) was filled with resin and light cured for 40 seconds under a light intensity of 1000 mW/cm2. The light cured specimens were then removed from the mold and aged for 24 hours in a 37℃ dark dry chamber. The aged specimens were then immersed in three pH solutions (pH 3, pH 6, and pH 9) for 14 days. Distilled water was mixed with dilute acetic acid to make the pH 6 and 3 solutions. The pH 9 solution was prepared by mixing dilute NaOH with distilled water. Mixing with acetic acid or NaOH was performed under slowly stirring conditions using a magnetic bar. All the processes were carried out at 23 ± 1℃ and 60 ± 3% relative humidity.
After immersion in the solutions at one of three pHs, each specimen was tested in three immersion beverages for 3 days: distilled water (DW); coffee (CF)-0.45 g of instant coffee (Taster’s Choice, Nestlé USA, Inc., Glendale, CA, USA) powder/50cc distilled water, without sugar or cream; and grape juice (GJ) (Fresh grape juice, Seoul Diary Co., Seoul, Korea).
2. Measurement of color
A spectrophotometer (CM-3600d, Konica Minolta, Osaka, Japan) was used to measure the color of the specimens. The initial color of the light-cured specimens was measured by placing the specimen at the center of the target mask in reflectance (%R) mode against a white and black background. The target mask had a 6 mm hole at the center. This hole enables consistency in specimen placement during the measurements. After the first color measurement, each specimen was immersed in the designated test beverage for 3 days. During immersion, the beverage solutions were replaced daily. After 3 days, the specimens were removed from the test solution and rinsed with running water. The remaining water was removed with tissue paper. The second color measurement using the immersed specimens was performed under the same measurement conditions as before. From the measured reflectance data, the CIEL*a*b* color values were evaluated using the internal software of the measurement system.
3. Calculation of color difference (ΔE*) and translucency parameter (TP)
The CIEL*a*b* color system is a three-dimensional color measurement system. The color difference (ΔE*) was obtained using the following formula:
| (1) |
where Δ is the difference between the first and second measurements. Here, L* refers to the lightness coordinate, which has a value ranging from 0 for perfect black to 100 for perfect white, and a* and b* are the chromaticity coordinates on the green–red (-a* = green; +a* = red) and blue–yellow (-b* = blue; +b* = yellow) axes, respectively.
The values of the translucency parameter (TP) were determined by calculating the color difference between the readings over a black and white background for the same specimen using the following equation:
| (2) |
where subscripts ‘B’ and ‘W’ refer to the color coordinates over a black and white background, respectively. The change in the translucency parameter, ΔTP (TP after immersion in test beverages minus TP before immersion in the test beverages), was calculated for each specimen.
4. Statistical analysis
The results of the color change were analyzed by two-way ANOVA for the test beverage and solution pH. A post hoc Tukey test was followed using a multiple comparison procedure. All tests were analyzed at p < 0.05.
Ⅲ. RESULTS
Tables 1 and 2 list the mean and standard deviations in the CIEL*a*b* color coordinates and color changes (ΔL*, Δa* and Δb*) of the composite resin after immersion in different staining beverages.
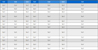
Mean and standard deviation values of color coordinates(ΔL*, Δa*, Δb*) after immersion in staining beverages for 3 days.
The ranges of ΔL* in the DW, CF and GJ groups were -0.18 ~ -0.48, -1.83 ~ -3.20 and -2.68 ~ -3.29, respectively. In the CF and GJ groups, ΔL* was higher than that in the DW group. The range of Δa* in all groups were -0.02 ~ 1.02. The change in Δa* was similar in all groups. The range of Δb* in the DW, CF and GJ groups were -0.18 ~ -0.23, 2.14 ~ 4.50 and 0.10 ~ 0.36, respectively. In the CF group, Δb* was higher than the others.
Table 3 lists the mean and standard deviations of the ΔE values of resin composite with different staining beverages after exposure to the three pH solutions. ΔE of the tested specimens was significantly different among three staining beverages examined (p < 0.05). On the other hand, no significant differences were observed between the pretreatments of the different pH solutions (p > 0.05).

Color changes (ΔE*) and standard deviations in the specimens of various pH pretreatment after 3 days immersion of staining beverage.
Staining is considered to be clinically unacceptable when the ΔE values are equal to or greater than 3.3. In this study, all ΔE values were nearly equal to greater than 3.3 except for the DW group. For distilled water, the color difference was only slight (ΔE; 0.30 ~ 0.64). The highest ΔE values were observed in the CF group at pH 3.
Table 4 shows the TP values and ΔTP before and after immersion in the staining beverages. The values of TP after immersion in the staining beverages decreased except for the DW group. The ΔTP values of the GJ group were higher than the others.
Ⅳ. DISCUSSION
The clinical use of composite resin as a restorative material has increased substantially in recent years because of the excellent esthetic properties and simplified bonding procedures (Lu et al., 2005). Despite the recent formulation improvements, the color stability of the composite resins after long term oral environment exposure is a concern in dental restorative procedures. An unacceptable color match is the primary reason for the replacement of composite resin restorations (Wilson et al., 1997).
Composite discoloration is a result of the interactions between the external colorants and composite resin materials. The adsorption of external colorants onto the surface and the absorption of such into the resin matrices can cause color changes and compromise the aesthetic outcomes (Ren et al., 2012). Despite this, the discoloration of composite resin can be affected by the acidic pH levels in the mouth. Therefore, the discoloration of resin affected by various pH conditions can be significantly different from that caused by only colorant absorption into the resin matrices.
Acidity in the oral environment can lead to the wear and surface degradation of composite restorations, resulting in unaesthetic external pigmentation, such as stains. Acidic media were reported to produce surface alterations in resin restorative materials (Turssi et al., 2002; Wongkhantee et al., 2006). A low pH can negatively affect the surface integrity by softening the matrix, causing a loss of structural ions and affecting the wear resistance of the dental materials (Asmussen, 1984; Damato et al., 1990; Oilo, 1992). On the other hand, Cilli et al. (Cilli et al., 2012) reported that alkaline media appears to be more suitable than acidic media in accelerating composite hydrolysis and producing microstructural damage. The strong influence of the alkaline medium on the composite properties was attributed to its interaction with OH ions during the hydrolysis process.
In the present study, the color stability of nanofilled resin composites was assessed under the actions of various pH solutions. All specimens were exposed to solutions at different pH before immersion in the staining beverages. Therefore, the low or high pH of exposed solutions might have affected the surface integrity of the material, thereby softening the matrix and increasing pigment absorption. On the other hand, in the present study, the pH of the exposed solutions had no effect on the color changes in the composite resin after immersion in the beverages.
Based on the attained data, the first null hypothesis tested in this study was accepted. Different pH levels before immersion in the staining beverages do not affect the color of the composite resin material. This might be because the pH used in this study was slightly acidic or alkaline, and might not have exerted much influence compared to a neutral medium. A similar result was reported in the literature (Ortengren et al., 2001a). According to Ortengren et al. (Ortengren et al., 2001a), they examined the influence of pH (4.0, 6.0 or 8.0) and storage time on the solubility and sorption of resin-based materials. On the other hand, they were not statistically significant for all materials. They concluded that the storage time was an important factor for the sorption and solubility behavior of the composite resin and the pH of the solution had little influence.
The pH of coffee and grape juice itself affected the composite resin surface, increasing pigment absorption. This effect was not considered in this study. Therefore, additional studies will be necessary.
A significant difference in discoloration was observed among the test beverages (P < 0.05). Therefore, the second null hypothesis of the present study was rejected. In the coffee group, ΔE was significantly higher than the others, which may be the larger changes in the L and b value than the others. This means that coffee induced a more yellowish color change in the composite resin than the other groups. In the grape juice group, the change in b value was not much larger than distilled water but the change in L value was larger. This suggests that the color change might be caused by the very dark sediments of grape juice on the composite surfaces. In other words, the composite resin was darkened more by grape juice.
Previous studies have shown that the greatest color change was observed when the materials were immersed in red wine (Patel et al., 2004; Bagheri et al., 2005; Ertas et al., 2006). On the other hand, they did not explain whether staining was caused by the alcohol or by the presence of pigments in the wine. To eliminate these variations, grape juice was used as an alternative beverage. Distilled water was used as a control in this study.
With regard to the severity of the color difference, several studies have shown that although the color differences of esthetic restorations showing ΔE > 1 are visually perceptible, they deemed ΔE > 3.3 to be the critical value for visual perception of the restoration (Schulze et al., 2003). In the present study, the ΔE values of coffee and grape juice (ΔE = 3.56 ~ 5.58, 3.01 ~ 3.44) ≥ 3.3 were considered clinically perceptible. Clinically, patients should be warned to expect composite resins to darken if they regularly drink staining beverages, such as coffee or grape juice. When the composite materials were immersed in distilled water, the color differences were imperceptible and clinically acceptable (ΔE = 0.30-0.64). This confirmed that water sorption itself did not alter the color of the composites to a considerable extent (Buchalla et al., 2002; Villalta et al., 2006).
In the case of the shade of TP, The values of TP after immersion into the staining beverages showed a decreasing tendency except for distilled water but the absolute values were low (0.40 ~ -2.52). ΔTP was higher in the grape juice group than the others, followed by coffee and distilled water group. This factor may darken the composite resin in the grape juice group and affect the TP value.
Under the results of the present study, one suggests that staining beverage solutions, such as coffee and grape juice, affected the color of the composite resin materials. From this study, coffee produced the most severe discoloration, whereas distilled water produced almost no perceptible discoloration, but a pretreatment with the pH solution did not affect the color of the composite resin materials. As the beverages tested in this study do not represent all substances to which restorative materials may be exposed to in the oral environment, and composite resin tested does not represent the materials to be restored in the oral cavity, additional studies will be necessary to examine the color stability of composite resins.
Ⅴ. CONCLUSION
Within the limitation of this study, ΔE of the tested specimens showed a significant difference among the staining beverages (p < 0.05) but there were no significant differences between the pretreatment of the pH solutions (p > 0.05). The values of TP after immersion in the staining beverages decreased except for distilled water, and ΔTP of grape juice was higher than the others. Overall, the color stability of the resin composite was not dependent on a pretreatment under pH conditions but was affected by exposure to a staining beverage.
Acknowledgments
This work was supported by a 2-Year Research Grant of Pusan National University
References
- Ardu, S., Braut, V., Gutemberg, D., Krejci, I., Dietschi, D., Feilzer, AJ., (2010), A long-term laboratory test on staining susceptibility of esthetic composite resin materials, Quintessence Int, 41, p695-702.
-
Asmussen, E., (1984), Softening of BISGMA-based polymers by ethanol and by organic acids of plaque, Scand J Dent Res, 92, p257-261.
[https://doi.org/10.1111/j.1600-0722.1984.tb00889.x]

-
Asmussen, E., Hansen, EK., (1986), Surface discoloration of restorative resins in relation to surface softening and oral hygiene, Scand J Dent Res, 94, p174-177.
[https://doi.org/10.1111/j.1600-0722.1986.tb01382.x]

-
Bagheri, R., Burrow, MF., Tyas, M., (2005), Influence of foodsimulating solutions and surface finish on susceptibility to staining of aesthetic restorative materials, J Dent, 33, p389-398.
[https://doi.org/10.1016/j.jdent.2004.10.018]

-
Barutcigil, C., Yıldız, M., (2012), Intrinsic and extrinsic discolouration of dimethacrylate and silorane based composites, J Dent, 40(Suppl 1), pe57-63.
[https://doi.org/10.1016/j.jdent.2011.12.017]

-
Buchalla, W., Attin, T., Hilgers, RD., Hellwig, E., (2002), The effect of water storage and light exposure on the color and translucency of a hybrid and a microfilled composite, J Prosthet Dent, 87, p264-270.
[https://doi.org/10.1067/mpr.2002.121743]

-
Cilli, R., Pereira, JC., Prakki, A., (2012), Properties of dental resins submitted to pH catalysed hydrolysis, J Dent, 40, p1144-1150.
[https://doi.org/10.1016/j.jdent.2012.09.012]

-
Damato, FA., Strang, R., Stephen, KW., (1990), Effect of fluoride concentration on remineralization of carious enamel: an in vitro pH-cycling study, Caries Res, 24, p174-180.
[https://doi.org/10.1159/000261262]

-
Ertas, E., Guler, AU., Yucel, AC., Koprulu, H., Guler, E., (2006), Color stability of resin composites after immersion in different drinks, Dent Mater J, 25, p371-376.
[https://doi.org/10.4012/dmj.25.371]

-
Fujita, M., Kawakami, S., Noda, M., Sano, H., (2006), Color change of newly developed esthetic restorative material immersed in food-simulating solutions, Dent Mater J, 25, p352-359.
[https://doi.org/10.4012/dmj.25.352]

-
Guler, AU., Yilmaz, F., Kulunk, T., Guler, E., Kurt, S., (2005), Effects of different drinks on stainability of resin composite provisional restorative materials, J Prosthet Dent, 94, p118-124.
[https://doi.org/10.1016/j.prosdent.2005.05.004]

-
Lee, W., Jung, JE., Jung, KH., Son, SA., Hur, B., Kwon, YH., Park, JK., (2014), The translucent and color change of nanofilled composite resin according to resin shades and staining solutions, Kor J Dent Mater, 41, p121-128.
[https://doi.org/10.14815/kjdm.2014.41.2.121]

-
Lu, H., Roeder, LB., Lei, L., Powers, JM., (2005), Effect of surface roughness on stain resistance of dental resin composites, J Esthet Restor Dent, 17, p102-108.
[https://doi.org/10.1111/j.1708-8240.2005.tb00094.x]

-
Millar, BJ., Robinson, PB., Inglis, AT., (1997), Clinical evaluation of an anterior hybrid composite resin over 8 years, Br Dent J, 182, p26-30.
[https://doi.org/10.1038/sj.bdj.4809289]

-
Minami, H., Hori, S., Kurashige, H., Murahara, S., Muraguchi, K., Minesaki, Y., Tanaka, T., (2007), Effects of thermal cycling on surface texture of restorative composite materials, Dent Mater J, 26, p316-322.
[https://doi.org/10.4012/dmj.26.316]

- Mohan, M., Shey, Z., Vaidyanathan, J., Vaidyanathan, TK., Munisamy, S., Janal, M., (2008), Color changes of restorative materials exposed in vitro to cola beverage, Pediatr Dent, 30, p309-316.
-
Mundim, FM., Garcia, LdFR. Pires-de-Souza FdCP., (2010), Effect of staining solutions and repolishing on color stability of direct composites, J Appl Oral Sci, 18, p249-254.
[https://doi.org/10.1590/S1678-77572010000300009]

- Oilo, G., (1992), Biodegradation of dental composites/glass ionomer cements, Adv Dent Res, 6, p50-54.
-
Ortengren, U., Andersson, F., Elgh, U., Terselius, B., Karlsson, S., (2001a), Influence of pH and storage time on the sorption and solubility behavior of three composite resin materials, J Dent, 29, p35-41.
[https://doi.org/10.1016/S0300-5712(00)00055-5]

-
Ortengren, U., Wellendorf, H., Karlsson, S., Ruyter, IE., (2001b), Sorption, solubility and identi®cation of substances released from composite materials in an aqueous environment, J Oral Rehabil, 28, p1106-1115.
[https://doi.org/10.1046/j.1365-2842.2001.00802.x]

-
Patel, SB., Gordan, VV., Barrett, AA., Shen, C., (2004), The effect of surface finishing and storage solutions on the color stability of resin-based composites, J Am Dent Assoc, 135, p587-594.
[https://doi.org/10.14219/jada.archive.2004.0246]

-
Ren, YF., Feng, L., Serban, D., Malmstrom, HS., (2012), Effects of common beverage colorants on color stability of dental composite resins: the utility of a thermocycling stain challenge model in vitro, J Dent, 40(Suppl 1), pe48-56.
[https://doi.org/10.1016/j.jdent.2012.04.017]

-
Ruyter, IE., (1995), Physical and chemical aspects related to substances released from polymer materials in an aqueous enviroment, Adv Dent Res, 9, p344-347.
[https://doi.org/10.1177/08959374950090040101]

-
Sarafianou, A., Iosifidou, S., Papadopoulos, T., Eliades, G., (2007), Color stability and degree of cure of direct composite restoratives after accelerated aging, Oper Dent, 32, p406-411.
[https://doi.org/10.2341/06-127]

-
Schulze, KA., Marshall, SJ., Gansky, SA., Marshall, GW., (2003), Color stability and hardness in dental composites after accelerated aging, Dent Mater, 19, p612-619.
[https://doi.org/10.1016/S0109-5641(03)00003-4]

-
Stober, T., Gilde, H., Lenz, P., (2001), Color stability of highly filled composite resin materials for facings, Dent Mater, 17, p87-94.
[https://doi.org/10.1016/S0109-5641(00)00065-8]

-
Turssi, CP., Hara, AT., Serra, MC., Rodrigues, AL. Jr, (2002), Effect of storage media upon the surface micromorphology of resin-based restorative materials, J Oral Rehabil, 29, p864-871.
[https://doi.org/10.1046/j.1365-2842.2002.00926.x]

-
Valinoti, AC., Neves, BG., Silva, EMd, Maia, LC., (2008), Surface degradation of composite resins by acidic medicines and ph-cycling, J Appl Oral Sci, 16, p257-265.
[https://doi.org/10.1590/S1678-77572008000400006]

- van Dijken, JW., (2001), Durability of new restorative materials in Class III cavities, J Adhes Dent, 3, p65-70.
-
Villalta, P., Lu, H., Okte, Z., Garcia-Godoy, F., Powers, JM., (2006), Effects of staining and bleaching on color change of dental composite resins, J Prosthet Dent, 95, p137-142.
[https://doi.org/10.1016/j.prosdent.2005.11.019]

- Wilson, NH., Burke, FJ., Mjor, IA., (1997), Reasons for placement and replacement of restorations of direct restorative materials by a selected group of practitioners in the United Kingdom, Quintessence Inter, 28, p245-248.
-
Wongkhantee, S., Patanapiradej, V., Maneenut, C., Tantbirojn, D., (2006), Effect of acidic food and drinks on surface hardness of enamel, dentine, and tooth-colored filling materials, J Dent, 34, p214-220.
[https://doi.org/10.1016/j.jdent.2005.06.003]

