Surface Wettability of Sandblasted Ti-alloy Discs: Ultraviolet (UV) Irradiation Effects Following Ethanol and Autoclave Pre-treatments
Abstract
Surface hydrophilicity of dental implants plays a pivotal role in promoting protein adsorption, cell adhesion, and ultimately osseointegration. Various surface treatments, such as sandblasting and SLA (sandblasted, large-grit, acid-etched), have been widely adopted to enhance hydrophilicity and ultraviolet (UV) irradiation has recently been applied for improved hydrophilicity. However, some studies have recently reported inconsistent hydrophilic outcomes following UV treatment. So, this study evaluated the surface wettability of Ti-alloy discs subjected to UV irradiation under different pre-treatment conditions. Sandblasted Ti-alloy discs were prepared and analyzed for surface morphology using scanning electron microscopy (SEM) and optical reflection techniques (machined and sandblasted). After surface sanitization with 70% ethanol and autoclaving of the sandblasted samples, they were exposed to UV light for 24 h and water contact angles were measured to investigate hydrophilicity. UV-treated groups exhibited increased hydrophobicity compared to untreated sandblasted controls. The group treated with 70% ethanol followed by UV exposure showed significantly higher water contact angles, indicating pronounced hydrophobicity. Even autoclaved samples displayed limited hydrophilicity, far from the level of hydrophilicity. These results suggested that airborne hydrocarbons and moisture-derived compounds, activated by UV light, rapidly adsorb onto rough Ti-alloy surfaces, reversing the intended hydrophilic effect. Pre-treatment conditions and post-UV irradiation were limited to achieve enhanced hydrophilicity based on the laboratory or general clinic environment. Therefore, effective packaging and surface protection protocols would be essentially required to prevent hydrocarbon contamination and ensure the stable formation of hydrophilic implant surfaces for clinical applications.
초록
치과용 임플란트의 표면 친수성은 단백질 흡착, 세포 부착을 촉진하고 궁극적으로 골융합을 유도하는 데 핵심적인 역할을 한다. 이러한 친수성을 향상시키기 위해 샌드블라스팅이나 SLA (sandblasted, large-grit, acid-etched)와 같은 다양한 표면 처리 방법이 널리 사용되어 왔으며, 최근에는 초친수성을 부여하기 위한 방법으로 자외선(UV) 조사도 적용되고 있다. 그러나 일부 연구에서는 UV 처리 후 일관되지 않은 친수성 결과가 보고된 바 있다. 이에 본 연구는 서로 다른 전처리 조건에서 UV 조사된 Ti 합금 디스크의 표면 습윤성을 평가하였다. 샌드블라스팅 처리된 Ti 합금 디스크를 제작하고, 주사전자현미경(SEM)과 광학 반사 분석을 통해 표면 형태를 관찰하였다. 이후 샌드블라스팅 시편을 70% 에탄올 소독 및 오토클레이브 처리한 뒤, 24시간 동안 UV에 노출시켰으며, 접촉각을 측정하여 친수성을 평가하였다. UV 처리군은 무처리 샌드블라스팅 대조군에 비해 오히려 소수성이 증가하였다. 특히, 70% 에탄올 처리 후 UV에 노출된 군은 접촉각이 유의미하게 증가하여 소수성을 나타냈다. 오토클레이브 처리군 역시 초친수성 수준에는 도달하지 못한 제한적인 친수성을 보였다. 이러한 결과는 UV 조사에 의해 활성화된 공기 중 탄화수소 및 수분 유래 성분이 거친 Ti 합금 표면에 빠르게 흡착되어, 의도한 친수성 효과가 반전될 가능성을 시사한다. 따라서 실험실이나 일반 임상 환경에서의 전처리 조건과 UV 처리만으로는 안정적인 초친수성을 확보하기 어려우며, 임상 적용을 위해서는 탄화수소 오염을 방지하고 친수성 표면을 안정적으로 유지할 수 있는 효과적인 포장 및 표면 보호 전략이 필수적으로 요구된다.
Keywords:
Titanium alloy, Sandblasting, Ultraviolet (UV) irradiation, Surface hydrophilicity키워드:
티타늄 합금, 샌드블라스팅, 자외서 조사, 표면 친수성Introduction
Dental implants have revolutionized the field of restorative dentistry, offering a reliable solution for replacing missing teeth. While natural teeth possess unique biological attributes, including proprioception and a periodontal ligament that allows for micro-movements, dental implants aim to mimic these functions through osseointegration (1). Osseointegration, defined as the direct structural and functional connection between ordered, living bone and the surface of a load-carrying implant, is crucial for implant stability and long-term clinical success (2). The process of osseointegration involves an initial interlocking between alveolar bone and the implant body, followed by biological fixation through continuous bone apposition and remodeling (3). This complex interaction is influenced by various factors, including implant material biocompatibility, surface properties, and the bone-implant interface (4). Titanium and its alloys have emerged as the materials of choice for dental implants due to their excellent biocompatibility and mechanical properties (5).
Natural teeth and dental implants differ significantly in their attachment mechanisms. Natural teeth are anchored to the jawbone through periodontal ligaments, which provide sensory feedback and allow for micro-movements (6). Dental implants achieve direct bone-to-implant contact without an intervening soft tissue layer, thereby relying exclusively on osseointegration for stability (7). To enhance osseointegration and improve clinical outcomes, various surface treatment techniques have been developed for dental implants (8). These techniques aim to modify the implant surface topography, chemistry, and energy, thereby influencing cellular responses and bone formation (9). Common surface modification methods include sandblasting, acid etching, plasma spraying, and anodization (8). More advanced techniques involve the creation of micro- and nano-scale surface features, which have shown promising results in promoting osteoblast adhesion, proliferation, and differentiation.
Hydrophilic implant surfaces facilitate protein adsorption in bioactive conformations, enhance the adhesion and spreading of osteogenic cells, and promote the stabilization of fibrin clots. This cascade of early biological events is vital for initiating new bone formation and accelerating osseointegration. Hydrophobic surfaces, on the other hand, tend to reduce the efficiency of protein–cell interactions, which may prolong bone healing and increase the risk of early implant failure. Conventional implant surface modifications have primarily focused on altering surface roughness by sandblasting, large-grit acid etching (SLA), anodization, and calcium phosphate coatings to create micro- and nano-topographies to improve mechanical interlocking with bone and provide favorable cues for osteoblast adhesion (10). Although these methods enhance bone–implant contact, many SLA or sandblasted exhibit hydrophobic characteristics when exposed to air, mainly due to spontaneous adsorption of hydrocarbons and airborne contaminants. Consequently, their effect on surface wettability remains inconsistent (11). UV irradiation (254 nm) could decompose organic hydrocarbons adsorbed on the titanium oxide surface and generates oxygen vacancies (12). Because this process restores the intrinsic surface energy of titanium which could convert hydrophobic surfaces into hydrophilic, numerous studies have reported that UV-treated implants exhibit superior protein adsorption, osteoblast adhesion, and enhanced osseointegration in animal and human studies. Moreover, UV photofunctionalization preserves the existing surface topography, allowing clinicians to maintain the benefits of roughened surfaces while improving their wettability (13, 14).
However, there remains debate regarding the reproducibility of UV-induced hydrophilicity under different pre-treatment or storage conditions. Some studies have reported inconsistent outcomes, with UV-treated surfaces displaying unexpected hydrophobicity (15). This study hypothesized that the hydrophilicity of sandblasted Ti-alloy (Ti-6Al-4V) surfaces could be reduced rather than enhanced when subjected to UV irradiation using a conventional UV sterilizer following sanitizing using 70% ethanol or autoclaving due to the counteraction by hydrocarbon or moisture-derived contamination.
Materials and Methods
1. Preparation of Titanium Alloy Discs
Titanium alloy (Ti-6Al-4V) discs were sectioned into discs measuring 10 mm in diameter and 5 mm in thickness. Discs were divided into two groups based on surface topography: machined smooth surfaces and sandblasted rough surfaces (Figure 1). For sandblasting, 50-µm alumina particles were projected at 0.25 MPa for 30 s at a fixed distance of 10 mm, followed by ultrasonic cleaning in deionized water for 10 m. then air-dried in a sterile environment.
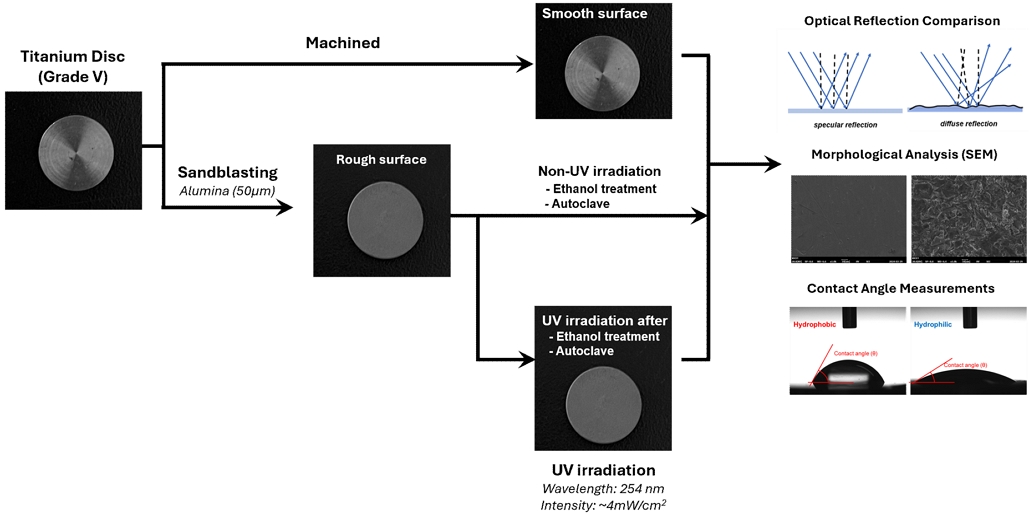
The procedure to analyze surface treatment effects with optical reflection, morphological difference, and contact angle measurements.
Ti-6Al-4V discs underwent two commonly applied sterilization protocols prior to UV treatment. For ethanol sterilization, the discs (nEtOH=5) were immersed in 70% ethanol for 1 h, gently rinsed with sterile deionized water. In parallel, the other samples(nautoclave=5) was sterilized by autoclaving, during which discs were sealed in sterilization pouches and exposed to saturated steam at 121℃ under 0.1 MPa for 15 m. Upon completion of either sterilization process, all discs were carefully handled with sterile forceps to prevent extraneous contamination. Both ethanol-treated and autoclaved groups were immediately transferred to the UV irradiation chamber, where a UV lamp emitting at 254 nm with an intensity of 4 mW/cm2 at a fixed distance of 50 cm was applied for 24 h.
2. Surface Characterization
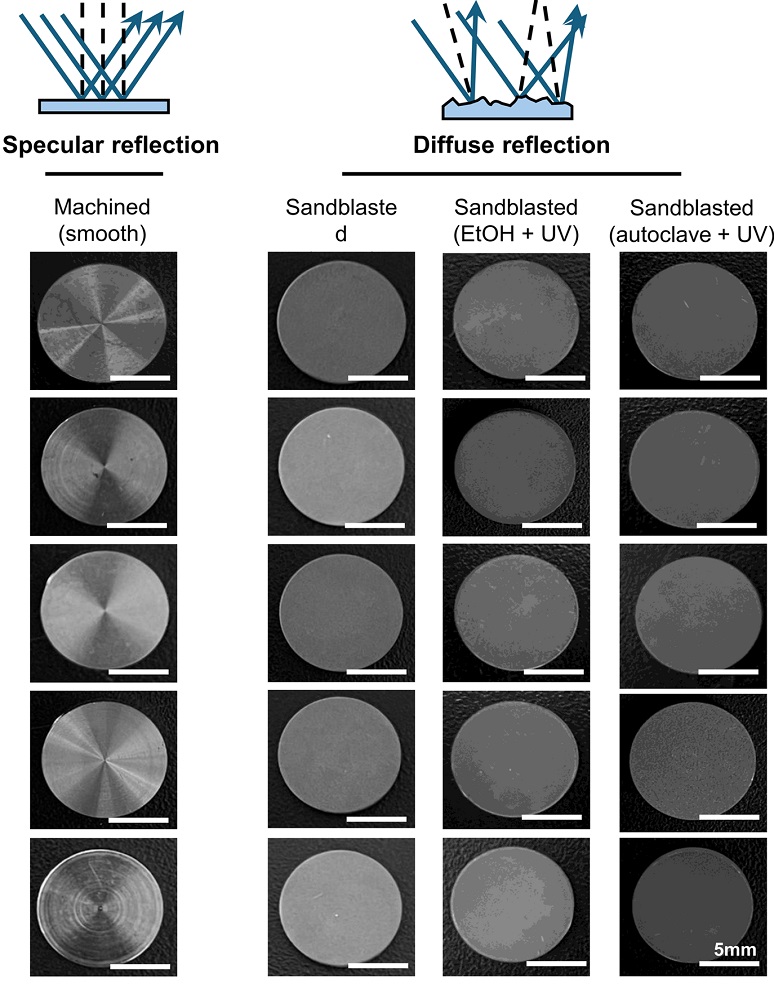
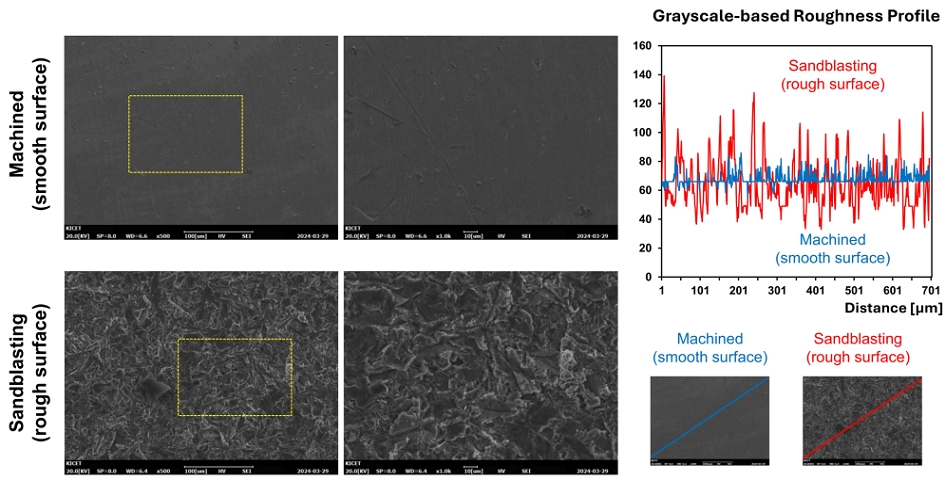
The surface properties of both groups were analyzed using multiple characterization techniques. The optical properties were evaluated by measuring reflection patterns. The machined surface demonstrated specular reflection characteristics, while the sandblasted surface exhibited diffuse reflection patterns, indicating differences in surface roughness. Scanning electron microscopy (SEM; EM-30, COXEM, Daejeon, Republic of Korea) was employed to examine the microscopic features of surfaces (SEtOH and Sautoclave) with 20.0 kV with a thin layer (~10 nm) of carbon coating to improve conductivity. High-resolution SEM images were captured and morphological characteristics were qualitatively compared between ethanol-treated and autoclaved surfaces
3. Contact Angle Measurement for hydrophilicity
Contact angle measurements were performed to evaluate the surface wettability characteristics. The hydrophobic and hydrophilic properties of both surfaces were assessed by measuring the contact angle (θ) of liquid droplets on the titanium surfaces under standardized conditions. A 10 µL droplet of deionized water was deposited onto the disc surface, and images were captured within 5 s. Temperature was 23.1±0.15℃ and humidity was 41.5±13.79% for the measurements. Hydrophilic surfaces were defined as having contact angles <20°, while hydrophobic surfaces had contact angles >70°.
4. Statistical Analysis
Data were expressed as mean ± SD. One-way ANOVA followed by Tukey’s post hoc test was applied to compare contact angles among groups. Significance was set at p<0.05. GraphPad Prism software was used for statistical and graphical analysis.
Results
1. Optical Reflection Properties
Smooth Ti-6Al-4V disc surfaces displayed specular reflection with visible concentric polishing lines and bright highlights but, sandblasted surfaces exhibited diffuse reflection, appearing matte with uniform light scattering. Ethanol treatment and autoclaving maintained these macroscopic reflection properties without noticeable alteration. UV irradiation also preserved the same reflection patterns, indicating that optical reflection characteristics were governed primarily by surface topography rather than surface chemistry (Figure 2).
2. SEM Morphology
SEM confirmed that machined discs showed smooth surfaces with faint polishing marks, while sandblasted discs displayed irregular microtopography with peaks and valleys. Ethanol treatment, autoclaving, and subsequent UV irradiation maintained these morphological features, with no observable alterations in microstructural characteristics (Figure 3).
3. Contact Angle Analysis
Machined smooth discs showed hydrophobicity (θMachined = 87.48±2.33°), while sandblasted discs displayed (θsandblasted = 28.36±11.65°). However, ethanol treatment prior to the UV treatment had increased contact angles (θEtOH+UV = 87.42±12.16°) to reach hydrophobicity and autoclaving before UV irradiation showed lower contact angle (θAutoclave + UV = 53.22±8.29°), higher hydrophilicity than EtOH + UV treatment (Figure 4). When UV irradiation for 1 h was applied after ethanol treatment or autoclaving, the treated specimens exhibited significantly reduced hydrophilicity compared with samples assessed immediately after sandblasting. Contrary to the expected outcome which UV irradiation could reliably induce hydrophilicity in sandblasted Ti-6Al-4V discs, pre-treatment with ethanol exacerbated hydrophobicity, and it was difficult to demonstrate autoclaving provided improvement (Figure 4A). To investigate the effect of UV exposure duration on surface wettability, additional groups with short-term (3 h) and long-term (24 h) UV irradiation were analyzed following ethanol or autoclave pretreatment (Figure 4B). When UV treatment was applied after ethanol pretreatment, both the 3-h (θEtOH + 3h-UV = 82.24 ±5.65°) and 24-h(θEtOH+24h-UV = 87.42±12.16°) exposure groups demonstrated significantly higher contact angles compared to the sandblasted-only group (θsandblasted = 28.36±11.65°). This trend was similarly observed in the autoclave-pretreated groups, where contact angles after 3-h (θAutoclave + 3h-UV = 57.16± 7.65°) and 24-h (θAutoclave + 24h-UV = 53.22±8.29°) UV exposure. Interestingly, prolonged UV exposure did not result in improved wettability and was associated with increased hydrophobicity in all tested conditions. These outcomes suggest that hydrocarbons and moisture-derived compounds may have been rapidly adsorbed onto rough titanium surfaces after UV exposure, thereby limiting the development of stable hydrophilic states.
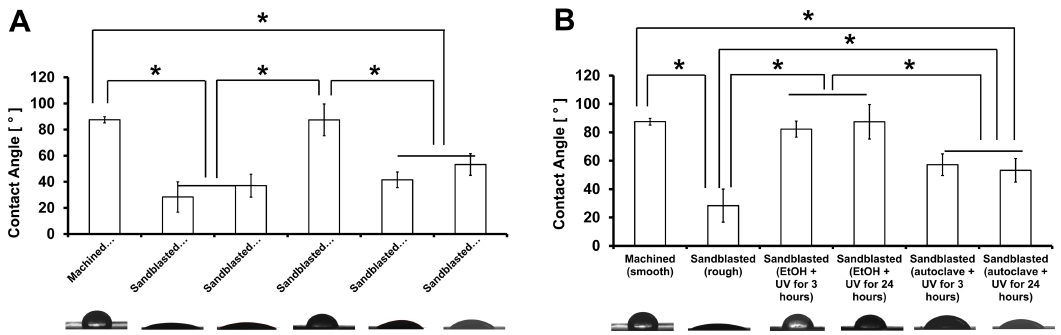
Figure. Surface wettability of titanium alloy discs evaluated by static contact angle measurements under various surface treatment conditions. (A) Comparison of contact angles among machined (smooth), sandblasted (rough), sandblasted (EtOH + dry or 24-h UV irradiation), and sandblasted (autoclave + dry or 24-h UV irradiation). (B) Contact angle analysis of sandblasted specimens after short-term (3 h) or long-term (24 h) UV irradiation, following ethanol or autoclave pretreatment. Data were represented as mean ± standard deviation (SD). Statistical significance was determined using One-way ANOVA followed by Tukey’s post hoc test with *p<0.05.
Discussion
The present study investigated the influence of UV irradiation on sandblasted Ti-6Al-4V discs subjected to ethanol sterilization or autoclaving. Although previous literature has consistently emphasized the ability of UV photofunctionalization to enhance surface hydrophilicity (13, 16), the current results demonstrated that surface wettability after UV treatment strongly depended on pre-treatment conditions. Ethanol-pretreated specimens displayed pronounced hydrophobicity following irradiation, and autoclaved specimens maintained only moderate hydrophilicity compared with sandblasted controls. These findings highlight the variability of UV-induced surface modification and the importance of environmental and procedural factors in determining the final surface state.
Earlier studies have described UV irradiation as a process capable of decomposing hydrocarbons, generating oxygen vacancies, and restoring titanium surface energy (17, 18). This cascade of changes has been associated with favorable biological responses, including improved protein adsorption, osteoblast attachment, and enhanced osseointegration (19, 20). However, the present results indicate that surface contamination or pre-treatment residues may significantly alter this process. The increase in contact angle observed in the ethanol + UV group suggests that residual organic compounds from ethanol processing may react under UV exposure, leading to stable hydrophobic films. Similarly, autoclaving introduced changes that limited the shift toward enhanced hydrophilicity, possibly due to deposition of hydrocarbons during steam sterilization. Other key factors could be the rapid adsorption of environmental compounds. Titanium surfaces exposed to UV irradiation acquire extremely high surface energy, which makes them highly reactive to airborne hydrocarbons and moisture-derived molecules (15, 20). On roughened sandblasted surfaces, with increased surface area and numerous microstructural valleys, such adsorption may occur rapidly and extensively.
From a clinical perspective, these findings underscore the importance of contamination control for implant surface management. Although UV treatment has been described as a simple chairside technique to improve implant wettability, the present data demonstrate that pre-treatment steps such as ethanol soaking or autoclaving can significantly influence the outcome. Reliance on UV irradiation alone may therefore lead to unpredictable surface states. Several strategies have been proposed to preserve hydrophilicity in clinical implants. One established approach involves storing implants in isotonic saline to prevent airborne hydrocarbon adsorption, as implemented in commercially available hydrophilic implant systems. Another potential solution could be the integration of UV activation with sealed packaging to protect freshly irradiated surfaces until the moment of clinical use. These approaches aim to minimize re-contamination and provide stable surface conditions favorable for osseointegration.
The present study has limitations that should be considered. First, the analysis was restricted to physicochemical characterization without biological validation. Protein adsorption, osteoblast adhesion, or in-vivo osseointegration assays would provide valuable evidence regarding the functional consequences of the observed changes in wettability. Second, the experimental design focused on Ti-6Al-4V discs under a single irradiation condition (254 nm for 24 h). Surface modifications or shorter irradiation durations may produce different outcomes. Finally, the relatively small sample size warrants cautious interpretation and further confirmation in larger studies.
Although only contact angle measurements were conducted in the present study, future work will include X-ray photoelectron spectroscopy (XPS) and time-of-flight secondary ion mass spectrometry (ToF-SIMS) analyses to clarify the extent of hydrocarbon decomposition and residual organic species on the UV-treated surfaces. Such chemical characterization will provide stronger evidence for the observed changes in wettability and surface reactivity. In addition, optimization of irradiation protocols, development of protective packaging systems, and integration of biological outcome measures will be essential for translating photofunctionalization into reliable clinical applications. In addition, a calvarial defects in animal models could provide in-vivo validation by pursuing 1) quantitative analysis of the protein attachment to the treated Ti-alloy surfaces for osseointegration by blood-derived proteins or osteoinductive proteins, 2) spatiotemporal assessments of subsequent bone tissue regeneration at the implant interface, and 3) evaluation of bone–implant interfacial adhesion through direct measurement of bone apposition and mechanical bonding strength. These investigations could allow to establish a more precise and clinically relevant UV treatment protocol to achieve and maintain hydrophilicity of dental implant surfaces.
Conclusion
This study evaluated the effects of ethanol treatment and autoclaving on the wettability of sandblasted Ti-6Al-4V surfaces exposed to UV irradiation. Freshly sandblasted surfaces displayed hydrophilic properties, whereas ethanol-pretreated specimens became distinctly hydrophobic after UV exposure, and autoclaved specimens exhibited intermediate wettability. These outcomes suggest that organic residues and moisture-derived compounds can rapidly adsorb onto titanium after irradiation, thereby counteracting the expected hydrophilic effect. The findings emphasize that UV photofunctionalization in routine laboratory or clinical environments requires strict control of pre-treatment and handling conditions. Reliable clinical application of this technique will depend on protective packaging systems and contamination-prevention strategies that preserve surface hydrophilicity until the point of use. Further research integrating chemical analysis and biological validation is essential to establish protocols that ensure consistent and stable enhancement of implant surface bioactivity.
References
-
Lang NP. Oral Implants: The Paradigm Shift in Restorative Dentistry. J Dent Res. 2019;98(12):1287-93.
[https://doi.org/10.1177/0022034519853574]

-
Barfeie A, Wilson J, Rees J. Implant surface characteristics and their effect on osseointegration. Br Dent J. 2015;218(5):E9.
[https://doi.org/10.1038/sj.bdj.2015.171]

-
Hasani-Sadrabadi MM, Pouraghaei S, Zahedi E, Sarrion P, Ishijima M, Dashtimoghadam E, et al. Antibacterial and Osteoinductive Implant Surface Using Layer-by-Layer Assembly. J Dent Res. 2021;100(10):1161-8.
[https://doi.org/10.1177/00220345211029185]

-
Smeets R, Stadlinger B, Schwarz F, Beck-Broichsitter B, Jung O, Precht C, et al. Impact of Dental Implant Surface Modifications on Osseointegration. Biomed Res Int. 2016;2016:6285620.
[https://doi.org/10.1155/2016/6285620]

-
Inchingolo AM, Malcangi G, Ferrante L, Del Vecchio G, Viapiano F, Inchingolo AD, et al. Surface Coatings of Dental Implants: A Review. J Funct Biomater. 2023;14(5).
[https://doi.org/10.3390/jfb14050287]

-
Chang HH, Yeh CL, Wang YL, Huang YC, Tsai SJ, Li YT, et al. Differences in the biomechanical behaviors of natural teeth and dental implants. Dent Mater. 2021;37(4):682-9.
[https://doi.org/10.1016/j.dental.2021.01.003]

-
Albrektsson T, Jansson T, Lekholm U. Osseointegrated dental implants. Dent Clin North Am. 1986;30(1):151-74.
[https://doi.org/10.1016/S0011-8532(22)02100-0]

-
Wang Z, Wang J, Wu R, Wei J. Construction of functional surfaces for dental implants to enhance osseointegration. Front Bioeng Biotechnol. 2023;11:1320307.
[https://doi.org/10.3389/fbioe.2023.1320307]

-
Kahm SH, Lee SH, Lim Y, Jeon HJ, Yun KI. Osseointegration of Dental Implants after Vacuum Plasma Surface Treatment In Vivo. J Funct Biomater. 2024;15(10).
[https://doi.org/10.3390/jfb15100278]

-
Mandracci P, Mussano F, Rivolo P, Carossa S. Surface Treatments and Functional Coatings for Biocompatibility Improvement and Bacterial Adhesion Reduction in Dental Implantology. Coatings. 2016; 6(1):7.
[https://doi.org/10.3390/coatings6010007]

-
Kligman S, Ren Z, Chung CH, Perillo MA, Chang YC, Koo H, et al. The Impact of Dental Implant Surface Modifications on Osseointegration and Biofilm Formation. J Clin Med. 2021;10(8).
[https://doi.org/10.3390/jcm10081641]

-
Ohtsu N, Masahashi N, Mizukoshi Y, Wagatsuma K. Hydrocarbon decomposition on a hydrophilic TiO2 surface by UV irradiation: spectral and quantitative analysis using in-situ XPS technique. Langmuir. 2009;25(19):11586-91.
[https://doi.org/10.1021/la901505m]

-
Chang LC. Clinical Applications of Photofunctionalization on Dental Implant Surfaces: A Narrative Review. J Clin Med. 2022;11(19).
[https://doi.org/10.3390/jcm11195823]

-
Ogawa T. Ultraviolet photofunctionalization of titanium implants. Int J Oral Maxillofac Implants. 2014;29(1):e95-102.
[https://doi.org/10.11607/jomi.te47]

-
Kido D, Komatsu K, Suzumura T, Matsuura T, Cheng J, Kim J, et al. Influence of Surface Contaminants and Hydrocarbon Pellicle on the Results of Wettability Measurements of Titanium. Int J Mol Sci. 2023;24(19).
[https://doi.org/10.3390/ijms241914688]

-
Tabuchi M, Hamajima K, Tanaka M, Sekiya T, Hirota M, Ogawa T. UV Light-Generated Superhydrophilicity of a Titanium Surface Enhances the Transfer, Diffusion and Adsorption of Osteogenic Factors from a Collagen Sponge. Int J Mol Sci. 2021;22(13).
[https://doi.org/10.3390/ijms22136811]

-
Naauman Z, Rajion ZAB, Maliha S, Hariy P, Muhammad QS, Noor HAR. Ultraviolet A and Ultraviolet C Light-Induced Reduction of Surface Hydrocarbons on Titanium Implants. Eur J Dent. 2019;13(1):114-8.
[https://doi.org/10.1055/s-0039-1688741]

-
Suzumura T, Matsuura T, Komatsu K, Ogawa T. A Novel High-Energy Vacuum Ultraviolet Light Photofunctionalization Approach for Decomposing Organic Molecules around Titanium. Int J Mol Sci. 2023;24(3).
[https://doi.org/10.3390/ijms24031978]

-
Roy M, Corti A, Dorocka-Bobkowska B, Pompella A. Positive Effects of UV-Photofunctionalization of Titanium Oxide Surfaces on the Survival and Differentiation of Osteogenic Precursor Cells-An In Vitro Study. J Funct Biomater. 2022;13(4).
[https://doi.org/10.3390/jfb13040265]

-
Sugita Y, Saruta J, Taniyama T, Kitajima H, Hirota M, Ikeda T, et al. UV-Pre-Treated and Protein-Adsorbed Titanium Implants Exhibit Enhanced Osteoconductivity. Int J Mol Sci. 2020;21(12).
[https://doi.org/10.3390/ijms21124194]